Silver Diamine Fluoride와 요오드화 칼륨 도포 후 변화하는 탈회 상아질의 내산성 평가
Evaluation of Acid Resistance of Demineralized Dentin after Silver Diamine Fluoride and Potassium Iodide Treatment
Article information
Abstract
본 연구는 Silver diamine fluoride (SDF)와 요오드화 칼륨(KI)이 이차 우식에 노출된 상아질의 내산성에 미치는 영향을 실험실 환경에서 평가하는 것이다. 소의 절치를 이용하여 상아질 시편을 제작한 뒤 인공적으로 초기 우식병소를 형성하여 음성 대조군, 양성 대조군, SDF 도포, SDF 도포 후 KI 도포(SDFKI)의 4개 군으로 나누었다. 음성 대조군을 제외한 각 군 당 4개의 시편 상면에 Streptococcus mutans, Lactobacillus casei 및 Candida albicans를 포함하는 다종 우식원성 세균을 접종하고 28일 간 배양하였다. 음성 대조군은 초기 우식병소 형성 뒤 인산 완충 용액에 담가 28일 간 보관하였다. 모든 시편의 탈회 정도는 micro-CT를 사용하여 분석하여 SDF와 KI가 탈회 상아질의 내산성에 미치는 영향을 비교, 평가하였다. 실험 결과 SDF와 SDFKI군에서는 양성 대조군에 비해 유의하게 탈회 깊이가 감소하여 SDF와 SDFKI의 도포가 탈회 상아질의 내산성을 증가시켜 이차 우식에 대한 저항성을 향상시킨 것을 확인할 수 있었다. KI 도포는 SDF의 항균 효과에 대해 유의한 영향을 미치지 않았다. 본 연구 결과 SDF와 SDFKI 도포는 탈회 상아질의 내산성을 증가시켜 이차 우식의 예방에 효과가 있었다. 또한 KI는 SDF의 항균 효과에 유의한 영향을 미치지 않으면서 검은 변색의 가능성을 줄일 수 있기 때문에 임상에서 효과적으로 사용될 수 있을 것으로 생각된다.
Trans Abstract
This study investigated the effects of silver diamine fluoride (SDF) and potassium iodide (KI) treatments on the acid resistance of dentin exposed to secondary caries. Sixteen bovine dentin specimens with artificially induced caries were assigned to the following four groups: untreated negative control, untreated positive control, SDF-treated (SDF), and SDF and KI-treated (SDFKI). Multispecies cariogenic biofilms containing Streptococcus mutans, Lactobacillus casei, and Candida albicans were cultured on the specimens for 28 days, except for the negative control group. Specimens from the negative control group were stored in phosphate-buffered saline for that period. After a cariogenic biofilm challenge, the degree of demineralization was evaluated using micro-computed tomography (micro-CT). As a result of data analysis using micro-CT, the demineralization depths of the negative control, positive control, SDF, and SDFKI groups were 149.0 ± 7 μm, 392.0 ± 11 μm, 206.0 ± 20 μm, and 230.0 ± 31 μm, respectively. The degree of demineralization was significantly reduced in the SDF and SDFKI groups compared with that in the untreated positive control group. There were no significant differences between the SDF and SDFKI groups. This study confirmed that SDF and SDFKI treatments increase the acid resistance of dentin to secondary caries. KI did not significantly affect the cariesarresting effect of the SDF.
Introduction
Dental caries is the most prevalent pediatric disease worldwide[1]. Although child dental health has remarkably improved compared to the past, significant proportions of low income, socially vulnerable, and minority children continue to be at risk of dental caries[2,3]. Although caries affects 60 - 90% of children[4], most caries lesions do not receive appropriate treatment for a variety of reasons, including the child’s anxiety, noncompliance, or financial issues.
Non-invasive caries management techniques, such as atraumatic restorative techniques, can be used effectively for untreated dental decays. The World Health Organization (WHO) actively promotes these for the treatment of dental caries in developing countries, and more recently, developed countries are increasingly accepting these techniques[1].
Silver diamine fluoride (SDF) application has been suggested as an alternate method of treating dental caries. SDF is a colorless alkaline liquid containing approximately 24 - 28% silver and 5 - 6% fluoride[5,6]. It has been used to prevent and arrest dental caries since the 1960s[7], and has been shown to be effective in arresting caries and inhibiting cariogenic biofilms formation in several studies. The application of SDF can be well accepted by children as it is simple, painless, and does not require local anesthesia or tooth drilling. However, the black discoloration that appears after the application of the SDF is the biggest drawback, making its clinical application difficult.
To overcome this problem, Knight et al.[8] proposed the innovative approach of applying a saturated potassium iodide (KI) solution just after SDF application. KI prevents discoloration by depositing excess silver ions as white silver iodide[9], without compromising the antimicrobial effect of SDF[10]. It would be ideal if KI could prevent the staining caused by SDF without weakening its efficacy in arresting and preventing caries.
In Korea, Riva star™ (SDI, Bayswater, Australia) is the first and only approved SDF product, and its domestic use started in June 2020. There are two agents in Riva star™. The first agent contains 30 - 35% SDF, and the second agent is a saturated KI solution[11]. The black discoloration that appears after the application of SDF can be reduced by immediately applying a sufficient amount of KI. Riva star™ was approved as a desensitizer, but it is widely used as an off-label for caries management of patients who have difficulty in dental treatment such as children or the disabled.
This study evaluated the effects of SDF and SDF followed by KI treatment on the acid resistance of demineralized dentin and, consequently, on the progression of secondary caries. Multispecies cariogenic biofilms were used to form secondary caries to reflect the actual oral environment as much as possible.
Materials and Methods
1. Specimen preparation
The specimen was prepared using an intact bovine incisor without caries, discoloration, or structural defects. The enamel of the bovine incisors was removed, and the dentin was cut into specimens of size 4.0 mm × 3.0 mm × 2.0 mm with a low-speed diamond disk under running water. The surface of the specimens was polished with a 1000 grit sand paper, and the side and rear surfaces were sealed with silicone (Examixfine injection type, GC Corporation, Tokyo, Japan) to assess the upper surface. Sixteen specimens were prepared and sterilized using ethylene oxide gas.
2. Artificial caries formation
A demineralization solution for inducing artificial caries was prepared as previously described[12]. The solution was then sterilized using a 0.22 μm vacuum filter system (JetBiofil, Guangzhou, China), and the specimens were immersed in the sterilized demineralization solution and stored for 7 days in a 37°C incubator to induce artificial caries.
3. Dentin specimen treatment
The Riva Star™ was applied to the specimens following the manufacturer’s instructions. The 16 specimens were divided into 4 groups: untreated negative control, untreated positive control, SDF-treated (SDF), and SDF and KI-treated (SDFKI). Using a sterile super-fine-sized microbrush, 1.0 μL of the first Riva Star™ agent was applied to the SDF group specimens for 10 seconds. In the SDFKI group, 1.0 μL of the second agent was applied for 10 seconds immediately after the application of the first agent. The second agent was administered twice to the specimens to ensure thorough application. After treatment, all specimens in the SDF and SDFKI groups were washed with sterile distilled water for 3 minutes.
4. Multispecies cariogenic biofilm challenge
The microorganisms used for the cariogenic challenge were Streptococcus mutans ATCC 25175, Lactobacillus casei ATCC 334, and Candida albicans ATCC 18804. Multispecies cariogenic biofilms were prepared based on the previous study[13]. The bacteria were cultivated on blood agar plates at 37°C for 24 hours until the isolated colonies were visible. The colonies were then transferred to brain heart infusion broth (BHI broth; Becton, Dickinson and Company, Sparks, MD, USA) and cultured at 37°C for 18 hours under aerobic conditions supplemented with 5% CO2.
All bacterial species were diluted to 1 × 109 colonyforming units (CFU)/mL using BHI broth for inoculation. A multispecies bacterial solution was prepared with 1940 μL of BHI broth containing 1% sucrose, and 20 μL of S. mutans, L. casei, and C. albicans respectively. All specimens, except for those in the negative control group, were immersed in this bacterial solution for 28 days in a 37°C CO2 incubator; the medium was replaced every 24 hours. Four specimens from the negative control group were stored in phosphate-buffered saline (PBS) under the same conditions. The treatment of the specimens from each group is shown (Table 1).
5. Assessment of demineralized lesions
Specimens were scanned using X-ray micro-computed tomography (SkyScan 1273; SkyScan, Antwerp, Belgium) to assess demineralized lesion depth. To reduce low energy radiation, a 1.0 mm thick aluminum filter was used. The scan was performed at a voltage of 90 kV and a current of 166 μA, with a spatial resolution of 12 μm. The scan results for each specimen were reconstructed using NRecon reconstruction software (SkyScan). After reconstruction, CTAn software (SkyScan) was used to view the images. The demineralization depth of each specimen was measured by dividing the specimen into vertical thirds, selecting the deepest demineralization point in the section, and averaging the total of 3 points. The measurement process using CTAn software is shown (Fig. 1).
6. Statistical analysis
IBM SPSS version 25.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. The Kruskal Wallis test, followed by Bonferroni post-hoc analysis, was performed for each group to verify statistical significance.
Result
A representative image of micro-CT from each group is shown (Fig. 2). The mean demineralization depths (± standard deviation [SD]) in the negative control, positive control, SDF, and SDFKI groups were 149 ± 7 μm, 392 ± 11 μm, 206 ± 20 μm, and 230 ± 30 μm, respectively (Table 2, Fig. 3). A statistically significant difference was detected between the negative control group and other groups (Table 3). SDF and SDFKI significantly reduced the degree of demineralization caused by multispecies biofilms. The demineralization depth was significantly reduced in the SDF and SDFKI groups compared to that in the positive control group. However, no statistically significant difference was observed between the SDF and SDFKI groups.

Demineralized area of each group obtained from micro-CT. (A) Negative control group, (B) Positive control group, (C) SDF group, (D) SDFKI group.
SDF = Silver diamine fluoride, SDFKI = Silver diamine fluoride and potassium iodide.
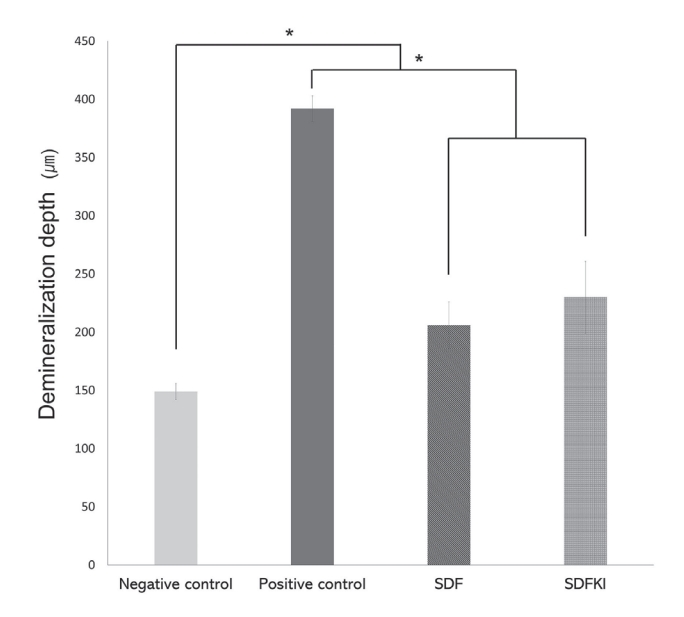
The mean and standard deviation of demineralization depth of the groups.
*: Means significant differences between the groups by Bonferroni’s post-hoc test.
SDF = Silver diamine fluoride, SDFKI = Silver diamine fluoride and potassium iodide.
Discussion
SDF can arrest existing carious lesions and prevent the formation of new lesions through multiple modes of action[1]. First, SDF forms materials that increase the resistance of peri-tubular and inter-tubular dentin to acid decalcification, and consequently retards the penetration of acids into the deeper layers of the dentin[14]. It is well known that silver ions have antimicrobial properties. Silver compounds have been used in dentistry for more than a century for caries management[15]. Fluoride forms fluorapatite, which increases dentin hardness, promotes remineralization, and prevents further demineralization of tooth structure. It also prevents caries owing to its bacteriostatic effects. Silver and fluoride ions penetrate 50 - 200 μm into the dentin. Yamaga et al.[16] suggested that the combination of fluoride and silver ions may prevent the loss of calcium and phosphate ions from teeth.
The second action is blocking the dentinal tubules to prevent the invasion of acids and cariogenic bacteria. The sensitivity of teeth can also be reduced by obturation[1]. In this study, a scanning electron microscope (SEM, JSM-IT500, JEOL, Tokyo, Japan) was additionally used to observe the surface of the specimens after micro-CT analysis (Fig. 4). While the clean dentinal tubules were observed in the SEM images of the specimens from the control groups, blocked dentinal tubules with particles produced by SDF and SDFKI application were observed in the SEM images of the specimens from SDF and SDFKI groups.
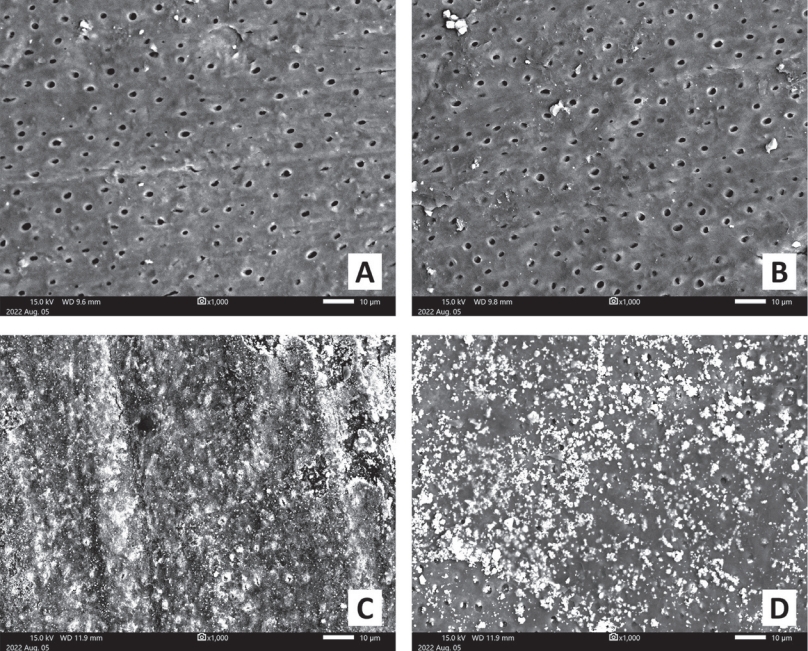
Scanning electron microscope images (× 1,000) of dentin specimen. (A) Negative control group, (B) Positive control group, (C) SDF group, (D) SDFKI group.
SDF = Silver diamine fluoride, SDFKI = Silver diamine fluoride and potassium iodide.
The third mechanism is the inhibition of the metabolism of cariogenic bacteria through anti-enzymatic action, which causes their death and reduces colonization. Mei et al.[17] reported that SDF inhibits the formation of multispecies cariogenic biofilms including S. mutans, Lactobacillus acidophilus and Actinomyces naeslundii. They also found that SDF reduced dentin demineralization.
Dental caries results from interactions between acidogenic bacteria and a variety of host factors, including teeth and saliva[18]. Owing to their acidogenic and aciduric characteristics, Streptococcus and Lactobacillus species contribute to the initiation and development of dental caries, respectively[19]. C. albicans is highly associated with early childhood caries[20] and decay-missingfilled (DMF) scores[21]. The microbiology of primary and secondary caries are similar[22]. For these reasons, these 3 types of bacteria, which are highly associated with dental caries, were selected to form multispecies cariogenic biofilms.
In this study, secondary caries progressed in multispecies cariogenic biofilms over 28 days, and micro-CT was used to measure the degree of demineralization. This is because micro-CT was thought to be the most intuitive way to confirm the degree of demineralization while causing the least damage to the specimen. As a result, SDF application increases the acid resistance of demineralized dentin and prevents the progression of secondary caries. The SDF and SDFKI groups showed significantly reduced demineralization depths compared to the positive control group. This finding is consistent with those of previous studies showing that SDF prevents the formation and progression of dental caries[23-26]. No significant differences were found between the SDF and SDFKI groups. This indicates that KI does not affect the cariesarresting effect of SDF. According to Kim et al.[27], applying KI immediately after SDF does not significantly affect the antibacterial action of SDF. This finding concurs with the result of this study. However, Turton et al.[28] reported that after 12 months of SDF and SDF + KI application, lower rates of caries arrest were observed in children who received KI after SDF than in those who received SDF alone. In this study, the demineralization depth was higher in the SDFKI group than in the SDF group, however, the difference was not statistically significant. This is probably due to the differences between the clinical situation and laboratory environment. In clinical situations, many other factors and interactions are involved in the occurrence of dental caries.
Owing to the black staining of SDF, it has not been used widely in the clinic despite its effective antimicrobial activity. The recently released Riva Star™ compensates for this drawback by reducing staining by applying KI. Several studies have shown that the application of KI is effective in reducing discoloration after SDF[29-31]. During this experiment, black discoloration did not also show up in the specimens with KI coating(Fig. 5). This study shows that applying KI immediately after SDF does not significantly influence the effect of SDF. Therefore, it can be used to minimize staining while maintaining the caries-arresting effect of SDF.

Color changes after SDF and SDFKI application. (A) SDF application, (B) SDFKI application.
SDF = Silver diamine fluoride, SDFKI = Silver diamine fluoride and potassium iodide.
One limitation of this study is that it was performed in vitro for a short period. Mature biofilms have more intricate structures and higher cell densities. In addition, numerous microorganisms interact with each other and transmit stimuli in the actual oral environment. Thus, the actual effects of SDF and SDFKI observed in this study may differ from those observed.
The second limitation was that it was not confirmed whether each specimen was demineralized to the same degree after the artificial caries formation. The degree of demineralization may differ for each tooth and for each part of the tooth, even when the same demineralization solution was used for the same period of time. However, if micro-CT had been used after inducing initial caries, specimens would have been contaminated. Sterilization would have been necessary again, which may have caused significant damage to the specimens. Also, it was confirmed that the 4 specimens of the negative control group, which only induced initial caries, had similar demineralization depth.
Another limitation was the small sample size with a large standard deviation of SDF and SDFKI groups. Further studies, including a large sample size in an environment comparable to the actual oral cavity, are needed.
Conclusion
In this study, SDF and SDFKI applications increased the acid resistance of dentin to secondary caries. KI application immediately after SDF did not significantly affect the caries-arresting effect of SDF. Therefore, KI can be used in combination with SDF to prevent secondary caries and reduce the possibility of discoloration caused by SDF. Few studies have examined the impact of SDF and KI application on secondary caries prevention. In addition, this study is thought to be significant in that it used multispecies cariogenic biofilms to reflect the oral environment.
Notes
Conflict of Interest
The authors have no potential conflicts of interest to disclose.




