유아기형 골화석증 환자의 치과적 관리 : 7년간의 경과 관찰
Dental Management in a Patient with Infantile Osteopetrosis : A Case Report with a 7-Year follow-up
Article information
Abstract
골화석증은 파골세포의 기능 장애 및 증가된 골 밀도를 보이는 질환으로 그 중 유아기형 골화석증은 심각한 유형이다. 전신의 골경화와 범혈구감소증, 두개 신경 협착, 높은 감염위험성, 두부와 안모의 변형 등 다양한 증상을 유발한다. 대부분의 유아기골화석증 환자는 발달 지연과 왜소증을 보이며, 조기에 사망에 이를 수 있다.
14개월의 여성 환아가 유전치부위에 초기 우식병소를 주소로 전남대학교 치과병원 소아치과에 내원하였다. 환아는 4세에 재내원 하였으며 interferon-gamma, erythropoietin 치료를 받고 있었다. 성장 지연, 골격 변형, 좁은 상악궁, 총생, 선천적 영구치 결손, 우식증을 보였다. 소아과 의사와 협진하여 예방적 항생제 투여와 진정요법 후 치과 치료를 진행하였다. 이후 감염이 발생한 다수 유구치를 발치 후 상악에 가철성 연성 의치를 이용하여 구강 재건(rehabilitation)을 시행하였다.
골화석증 환자의 경우, 저하된 면역기능으로 인해 감염에 매우 취약하며, 출혈이나 발치와 연관된 골수염이나 패혈증이 유발 될 수 있으므로 소아과 의료진의 협조와 예방적 항생제의 사용에 관한 고려가 간단한 치과시술 시에도 필수적이다. 또한, 당분섭취 제한 및 구강위생관리를 위한 의료진의 적극적인 개입이 필요하다.
Trans Abstract
Osteopetrosis is characterized by impaired osteoclast function and increased bone density. Infantile osteopetrosis is a severe form of the disease and has characteristics such as diffusely sclerotic skeleton, pancytopenia, cranial nerve entrapment, infection susceptibility, and abnormal craniofacial appearance. Patients with infantile osteopetrosis often experience developmental delay, and may have a short life span.
A 14-month-old girl with osteopetrosis presented to the department of pediatric dentistry. Incipient caries on deciduous incisors were observed. The patient revisited 4 years of age. Besides medical problems, oral complications such as growth retardation, narrow upper arch, crowding, dental caries, and abnormal tooth development were observed. After consultation with her pediatrician, dental treatments were performed on the deciduous molars under sedation after a prophylactic antibiotic injection. At a periodic follow-up, multiple deciduous teeth were treated and extracted, and oralrehabilitation with a removable partial denture was initiated.
Patient with osteopetrosis are highly susceptible to infection because of their compromised immune system and problems associated with wound healing that lead to osteomyelitis or sepsis development.
Active participation in dental care for sugar intake management and proper oral hygiene are obligatory.
Ⅰ. Introduction
Osteopetrosis is a family of bone diseases and is characterized by osteoclast failure and impaired bone resorption[1]. It was first identified in 1904 by Albers-Schönberg and was described as “marble bone disease” because of intense bone sclerosis[1-2]. It comprises a clinically and genetically heterogeneous group of conditions that share the hallmarks of increased bone density on radiographs, and mutations in at least 10 genes have been identified as causative in humans[3,4].
The diagnosis of osteopetrosis is clinical and largely depends on the skeletal radiographic features[4]. Radiographic features of the disease include widespread sclerosis that affects the skull, spine, pelvis, and appendicular with defect in metaphyseal remodeling. The radiographic distinction between the cortical bone and cancellous bone disappears. In addition, computed tomography can be used to diagnose of the disease or to assess the auditory and optic canal. Genetic and laboratory testing can also be used to confirm the diagnosis of osteopetrosis[5-7].
Osteopetrosis is rare, and the estimating its overall incidence is difficult. Autosomal recessive osteopetrosis has an incidence of 1 in 250,000 infants, with a particularly high incidence rate reported in Costa Rica (3.4:100,000)[8].
The severity of osteopetrosis range from asymptomatic to infant fatality. The most severe forms tend to have an autosomal recessive inheritance, whereas the mildest forms are observed in adults and are inherited in an autosomal dominant manner[4]. Three clinically distinct forms of osteopetrosis, namely infantile, intermediate, and adult onset, have been identified based on age and clinical features[9].
Infantile osteopetrosis is a severe form of the disease and presents with diffusely sclerotic skeleton. Initial signs often include monocyclic anemia with hepatosplenomegaly owing to compensatory extramedullary hematopoiesis and increased susceptibility to infections because of granulocytopenia. If untreated, death owing to hemorrhage, pneumonia, severe anemia, or sepsis often occur during the first decade. Intermediate osteopetrosis is a less severe variant of infantile osteopetrosis[10]. Adult osteopetrosis is usually diagnosed later in life and has a less severe manifestation[11,12]. Osteopetrosis causes various oral complications such as deformation of jaws and teeth, increased susceptibility to infections, and dental caries[4,13,14]. Among these, Infantile osteopetrosis shows the most serious systemic and oral symptoms.
This study aimed to describe the dental management of a patient with a 7-years history of infantile osteopetrosis.
Ⅱ. Case Report
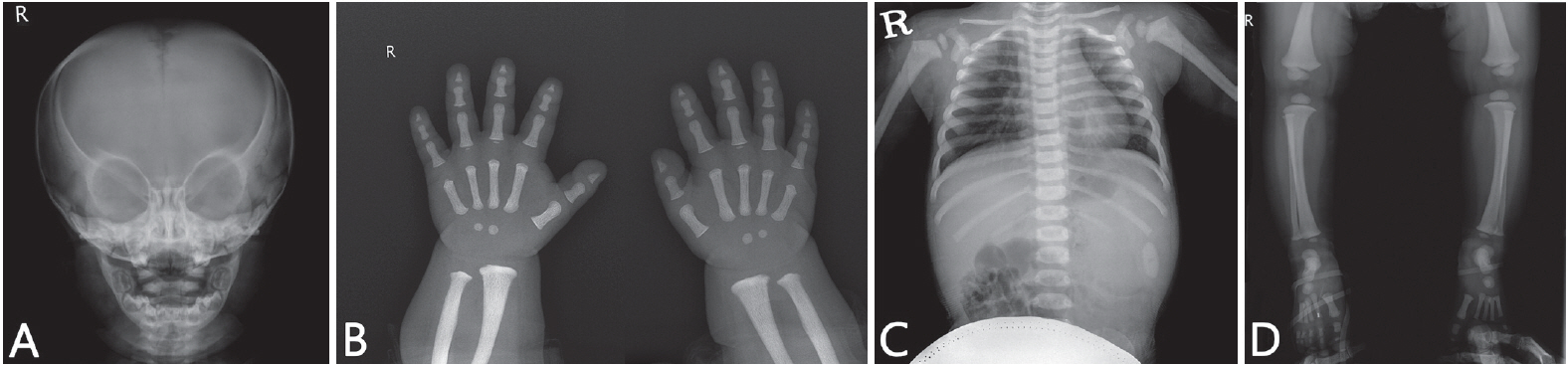
A 14-month-old girl with osteopetrosis was referred to the department of pediatric dentistry, Chonnam national university dental hospital for dental evaluation and treatment. Her chief complaint was multiple carious lesions. She was diagnosed with infantile osteopetrosis based on radiographic and laboratory examinations conducted at 8 months of age. She had splenohepatomegaly and pancytopenia (Fig. 1, Table 1).
The patient was systematically treated with interferongamma (Intermax Gamma®, LG Life Science Co., Korea) and erythropoietin (Nesp®, Kyowa Hakko Kirin Co., Japan). However, bone marrow transplantation and blood transfusion were not performed on religious grounds.
Clinical examination revealed incipient dental caries on the maxillary deciduous anterior teeth. Oral hygiene management was initiated considering systematic conditions. The parents were also instructed to perform thorough home dental care and periodic clinical examinations.
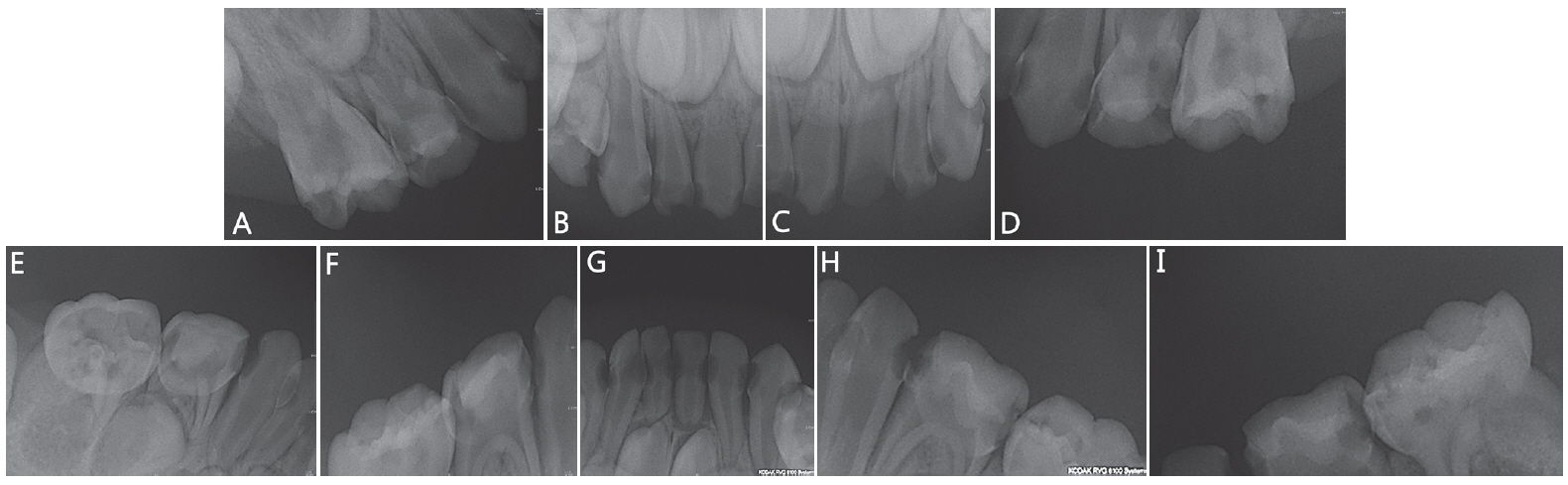
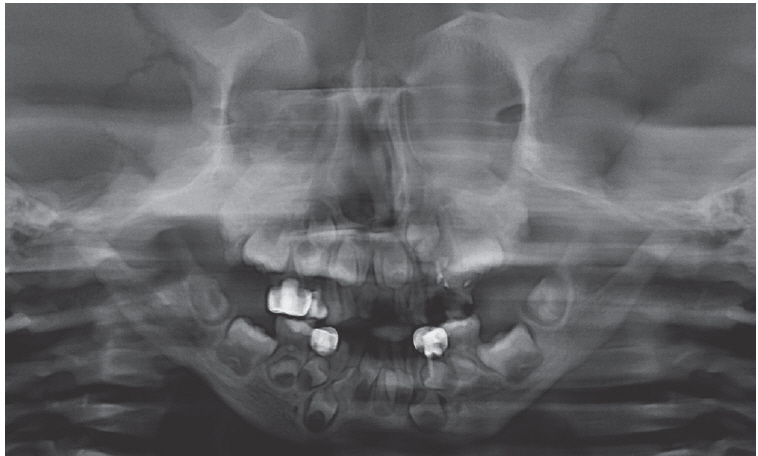
At a later visit at 4 years of age, the parents explained that they could not keep appointments due to patient’s systemic condition. The patient had a short stature, macrocephaly, exophthalmos, and clubbing on fingers (Fig. 2, Table 2). A panoramic radiograph revealed overall sclerosis of the facial and vertebral bone, developmental delay of the alveolar bone, short mandibular condyle, loss of mandibular angle, and congenitally missing the maxillary first premolars (Fig. 3). All primary teeth were completely erupted, and multiple caries on deciduous molars were observed. Periapical radiographic images revealed dental caries were observed in all primary teeth. In addition, periapical infection was observed on all deciduous first molars and the right upper second molar (Fig. 4). The extraction of teeth with periapical radiolucency for severe caries was initially recommended. However, a conservative treatment approach was planned because the parents were unwilling to approve a more aggressive treatment and the patient’s young age.
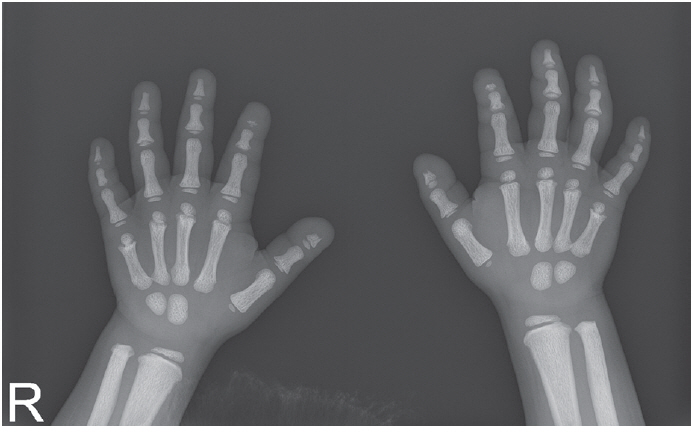
Bone resorption in the distal phalanx of thumb and index fingers and generalized bone density increase were noted on hand radiograph.
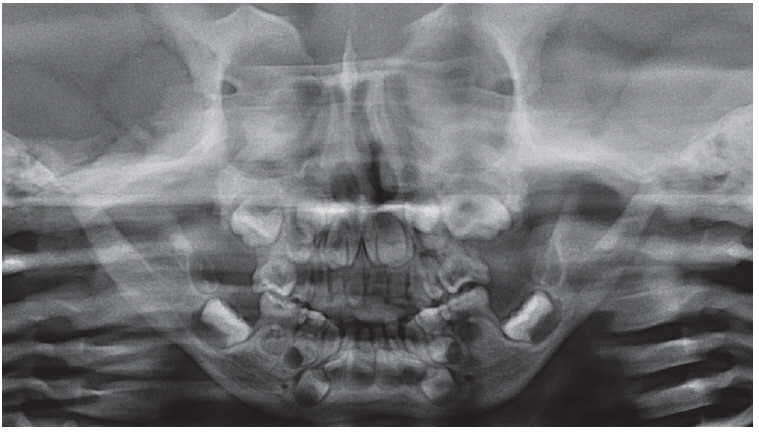
Panoramic radiograph of the patient at the age of 4. Congenital missing of maxillary first premolar, short mandibular condyle, growth retardation of maxilla and mandible were noted. Overall sclerosis of the facial and vertebral bone was showed.
Because the lack of the patient’s cooperation, a sedative treatment using Pocral®, Ucerax®, and nitrous oxide/oxygen (N2O/O2) inhalation was planned. The patient’s systemic condition and availability of therapeutic approaches such as cautions in using sedative medication and necessity for prophylatic antibiotics were discussed with the pediatrician. A prophylatic antibiotic injection (Augmentin®; Amoxicillin 20 mg, Clavulanate K 5 ml/kg/day) was administered. Endodontic treatment was performed on first deciduous molars and maxillary second deciduous molars. Restorative treatment was performed on all deciduous molars. During the procedures, oxygen concentration were fixed at 100% (5 L/min) because the functional arterial oxygen saturation (SpO2) intermittently dropped to 75 - 82% during N2O/O2 inhalation.
One year later, the patient revisited because of pain in the upper primary molars. Clinical and radiographic examinations revealed periapical radiolucency on the upper right first primary molar, and retained roots of the left primary molars (Fig. 5, 6). After consulting the pediatrician, #54, #64, and #65 were extracted after prophylactic antibiotics.
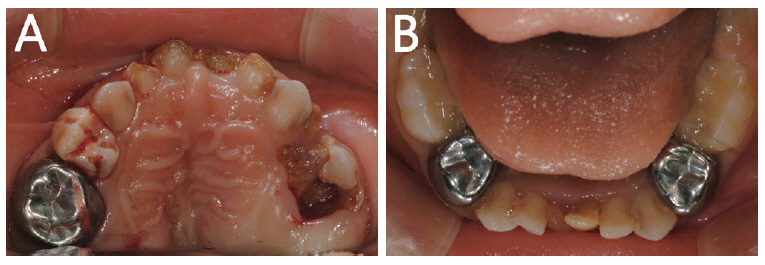
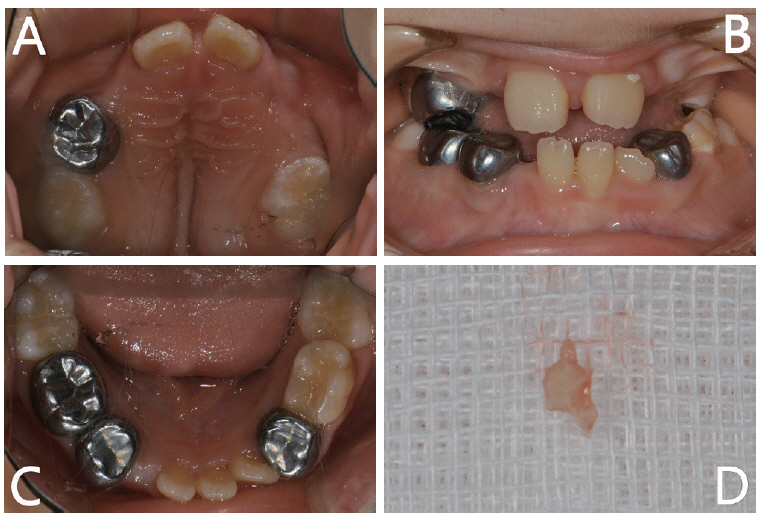
Intraoral photographs show multiple dental caries in the narrow upper arch. Anterior crowding of maxillary and mandibular arch is also noted (A, B).
At 6 years of age, patient had aesthetic and functional problems. Placement of removable prosthesis was planned to replace the extracted primary teeth until the eruption of the permanent teeth. According to the proper jaw relations, the maxillary flexible denture was fabricated and delivered to the patient (Fig. 7). A regular 3-month recall protocol was maintained to monitor the growth of the dental arches and eruption status of the permanent teeth.
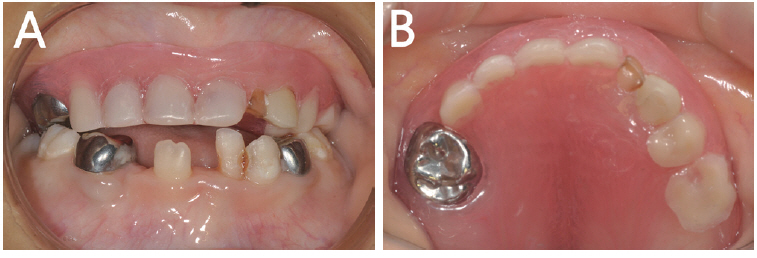
Removable flexible denture was fabricated to replace extracted primary teeth until eruption of permanent teeth (A, B).
At a regular follow-up visit, the eruption of permanent first molars was evident and sequestrums were also observed on the occlusal surfaces (Fig. 8, 9). As preventive management, occlusal sealant on the permanent first molars was applied. Periodic oral exam were planned.
Ⅲ. Discussion
Infantile osteopetrosis is a life-threatening condition and generally begins in utero. It presents at birth or within the first year of life. Patients with infantile osteopetrosis who survive childbirth might experience bone marrow failure, cranial nerve entrapment, general failure to thrive or characteristic faces[15].
Bone marrow failure causes pancytopenia with hepatosplenomegaly owing to compensatory extramedullary hematopoiesis. Increased susceptibility to infections are also caused by granulocytopenia[16]. To minimize the risk of medical complications, patients with hemoglobin levels less than 11 g/dL or oxygen saturation below 91% are required to be on hold for routine treatment until their health status improves. Patient with neutropenic status should not undergo invasive dental procedures without specialized pretreatment and precautions. After the necessary treatment is administered by the patient’s physician, the use of prophylactic antibiotics is dictated based on white blood cell and neutrophil counts. Prophylactic antibiotics are often recommended if the white blood cell count is less than 2,000 before initiating surgical procedures[17]. Anemia could cause acidosis, hypoxemia, dehydration, venous stasis, and hypothermia. Patients with the disease also undergo corticosteroid therapy. Thus, supplemental oxygen, adequate pain control and hydration, as well as active treatment of infection are recommended during dental procedures for patients with osteopetrosis[18].
Neurological problems such as optic atrophy, nystagmus and blindness, deafness and facial paralysis could develop because failure in remodeling of skull bones causes narrowing of the skull foramina and elevated pressure on various cranial nerves. Hearing loss is estimated to affect 78% of individuals with this type of disease. Nasal stiffness owing to the malformation of mastoid and paranasal sinuses can also occur[19-21].
The common orofacial findings in infantile osteopetrosis include facial deformity along with macrocephaly, hypertelorism, frontal bossing, and snub nose[1,4-6]. The patient in this case also exhibited such characteristic facial features. A panoramic radiograph revealed overall sclerosis of the facial and vertebral bones, developmental delay of the alveolar bone, and short mandibular condyle.
Common dental findings that accompany osteopetrosis include tooth eruption defects, hypodontia, enamel hypoplasia, abnormal pulp chamber and severe dental caries. Thickened lamina dura, early dental loss, and osteomyelitis associated with extraction can also develop[1,4,6,7,14].
Tooth eruption is critically dependent on osteoclast activity in the jaw to create a path for developing teeth through the alveolar bone to the oral cavity. Therefore, a characteristic feature of osteopetrosis is the absence or severe delay in tooth eruption[22].
Dental changes are probably the result of local environmental factors that affect the nutritional and spatial requirements of developing tooth germs. The results of reduced calcium and phosphorus ratio in both enamel and dentin suggest altered mineral metabolism in these tissues. These changes might also result in reduced hydroxyapatite crystal formation[1,23,24].
In this case report, refusal of the recommended treatment by the parents and limitations because of the patient’s age led to conservative treatment over simple extraction, however, some teeth that underwent root canal treatment were extracted after 1 year because of infection.
In patients with compromised immunity, as seen in those with infantile osteopetrosis, extraction could be a suitable option for apically infected teeth with uncertain prognosis. Because the vascular supply to the jaws and wound healing are compromised in patients with osteopetrosis, avascular necrosis and infection after dental extractions may lead to osteomyelitis[14]. Thus, the use of prophylactic antibiotics should be considered by the practitioner before dental treatment.
In this case, the patient’s SpO2 level intermittently decreased to 75 - 82% under sedation. After ceasing N2O/O2 inhalation and supplying 100% O2 inhalation, the patient’s SpO2 level stabilized. However, the decreased SpO2 level might have resulted from the patient’s systemic condition. Upper respiratory tract function might be drastically altered in children with infantile osteopetrosis[25]. Napoleon et al.[26] analyzed perioperative complications in the anesthetic management of 65 children with osteopetrosis. Major complications include airway difficulty (17.74%), inability to intubate (14.52%), and postoperative death (3.23%). Complication rates were higher than those for other pediatric patients. Thus, a practitioner should perceive the patients’ anatomic alteration of respiratory tract while planning a sedative treatment. In this case, the practitioner was particularly careful to avoid airway obstruction during procedures.
Hematopoietic stem cell transplantation (HSCT) is currently the only definitive medical treatment for severe osteopetrosis[4]. Bone marrow transplantation induces normalization of osteoclast function, which is a prerequisite for normal dental development and eruption of teeth. Jalevik et al.[27] reported that dental development and eruption of teeth can be reconstituted in a child with infantile osteopetrosis after early bone marrow transplantation. However, an appropriately matched donor is available for only about one-half of affected patients, and successful engraftment occurs in only approximately 45% of those undergoing bone marrow transplantation[28]. Furthermore, there are patients who cannot undergo HSCT owing to the high cost or religious grounds. Thus, various medical interventions, including corticosteroid, high-dose calcitriol, and interferon-gamma (IFN-γ), have been attempted[5,29,30]. Kim et al.[31] observed the subcutaneous IFN-γ injection could help to manage complications, delay disease progression, and prolong survival in patients with severe osteopetrosis[10,19]. The patient in this case was treated with long-term IFN-γ instead of blood transfusion or HSCT as a supportive treatment.
Based upon discussed findings, the practitioner should completely understand the patient’s condition and the following recommendations.
First, A sufficient explanation of tooth and jaw deformation should be given to the parents, and periodic examination of the patient should be performed.
Patient with Osteopetrosis have reduced immune function and experience bleeding and hindered wound healing ability. This could progress to osteomyelitis or sepsis. A cooperative approach with a pediatrician is necessary for every dental appointment. The use of antibiotics was considered for invasive treatment. Dental management of the patient with osteopetrosis requires a comprehensive approach to characteristic clinical problems, including hematological and metabolic abnormalities, recurrent infections, bone complications, and neurological sequela.
Above all, the primary objective of dental management for patient with osteopetrosis is to maintain healthy oral conditions through periodic oral hygiene management to prevent caries, infection, and early loss of teeth. Patient with infantile osteopetrosis are in a high-risk group for dental caries owing to the weakened dental structure, prolonged hospitalization, insufficient masticatory function due to liquid diet, and poor oral hygiene. Therefore, periodic clinical and radiographic examinations should be performed. Thorough oral hygiene management and restricted sugar intake should also be practiced.
This study presents a longitudinal observation of a person, and there may be limitations in complete representation of infantile osteopetrosis. Nevertheless, this case report might be helpful to dental practitioner to improve their understanding of dental care for patients with infantile osteopetrosis to provide the optimal oral health care.
Ⅳ. Summary
Infantile osteopetrosis is the most severe form of osteopetrosis and is characterized by diffusely sclerotic skeleton, monocyclic anemia, developmental delay, and infection susceptibility. Oral manifestations of the disease are delayed tooth eruption, congenial missing teeth, enamel hypoplasia and tooth decay. The use of prophylactic antibiotics and consultation with pediatricians are essential for invasive therapy.
The findings of this study emphasize the importance of preventive treatment and long-term follow-up for patient with osteopetrosis who are highly susceptible to dental caries.