|
 |
| J Korean Acad Pediatr Dent > Volume 46(3); 2019 > Article |
|
ņ┤łļĪØ
ņØ┤ ņŚ░ĻĄ¼ņØś ļ¬®ņĀüņØĆ ļ░£ņ╣śĒĢ£ ļ¦żļ│Ą Ļ│╝ņ×ēņ╣śņŚÉņä£ ņ¢╗ņØĆ ņ╣śņłś ņ£Āļל ņäĖĒżņØś ņżäĻĖ░ņäĖĒż ĒŖ╣ņä▒ņØä ņ£ĀņäĖĒż ļČäņäØņØä ĒåĄĒĢ┤ ņĢīņĢäļ│┤ļŖö Ļ▓āņØ┤ļŗż. 15ļ¬ģņØś ĒÖśņ×ÉļĪ£ļČĆĒä░ ņ▒äņĘ©ĒĢ£ ņĀĢņżæ Ļ│╝ņ×ēņ╣śņØś ņ╣śņłś ņäĖĒżļź╝ Ļ│äļīĆ ļ░░ņ¢æĒĢśņśĆĻ│Ā, 3Ļ│äļīĆņÖĆ 10Ļ│äļīĆ ņäĖĒżļōżņØä ņ£ĀņäĖĒż ļČäņäØņØä ņØ┤ņÜ®ĒĢśņŚ¼ ļČäņäØĒĢśņśĆļŗż. Ļ░äņŚĮņä▒ ņżäĻĖ░ņäĖĒż Ēæ£ņ¦Ćņ×É Ļ┤Ćņ░░ņŚÉ ņé¼ņÜ®ļÉ£ ĒĢŁņ▓┤ Ēæ£ņ¦Ćņ×É(CD)ļŖö ņ¢æņä▒ Ēæ£ņ¦Ćņ×ÉļĪ£ļŖö CD 73, CD 90, ĻĘĖļ”¼Ļ│Ā STRO-1 ņÖĆ ņØīņä▒ Ēæ£ņ¦Ćņ×ÉļĪ£ļŖö CD 34, CD 45ņØ┤ņŚłļŗż. 3Ļ│äļīĆņŚÉņä£ CD 73, CD 90ņØĆ Ļ░üĻ░ü 94.82%, 98.86%ņØś ņ¢æņä▒ļ░śņØæņØä, CD 34, CD 45ļŖö Ļ░üĻ░ü 2.25%, 2.52%ļĪ£ ņØīņä▒ ļ░śņØæņØä ļ│┤ņśĆņ£╝ļ®░, STRO-1ņØĆ 20.93%ļź╝ ļéśĒāĆļāłļŗż. 10Ļ│äļīĆņŚÉņä£ļŖö CD 73, CD 90ņØĆ Ļ░üĻ░ü 96.62%, 98.61%ņØś ļ░£ĒśäņØä ļ│┤ņśĆņ¦Ćļ¦ī, CD 34, CD 45ļŖö Ļ░üĻ░ü 3.86%, 4.14%ļź╝ ļéśĒāĆļāłļŗż. STRO-1ņØĆ 35.62%ļĪ£ ļ░£ĒśäļÉśņŚłļŗż. ņØ┤ņāüņØś Ļ▓░Ļ│╝ņŚÉņä£ Ļ│╝ņ×ēņ╣ś ņ╣śņłś ņ£Āļל ņäĖĒżļŖö Ļ░äņŚĮņä▒ ņżäĻĖ░ņäĖĒżņØś ĒŖ╣ņä▒ņØä Ļ░Ćņ¦Ćļ®░, 3Ļ│äļīĆņÖĆ 10Ļ│äļīĆ ļ¬©ļæÉņŚÉņä£ Ļ░äņŚĮņä▒ ņżäĻĖ░ņäĖĒżņØś ĒŖ╣ņä▒ņØä ņ£Āņ¦ĆĒĢśĻ│Ā ņ׳ļŗżĻ│Ā ņé¼ļŻīļÉ£ļŗż. ņØ┤ņŚÉ ļ╣ĀļźĖ ņä▒ņן ņåŹļÅäņÖĆ ļŖ”ņØĆ Ļ│äļīĆĻ╣īņ¦Ć ņ£Āņ¦ĆļÉśļŖö ņżäĻĖ░ņäĖĒżļŖźņØä Ļ│ĀļĀżĒĢĀ ļĢī, ņ╣śņĢä ņ£Āļל ņżäĻĖ░ņäĖĒżņØś Ļ│ĄņŚ¼ļČĆļĪ£ņä£ ļ¦żļ│Ą Ļ│╝ņ×ēņ╣śņØś ņČ®ļČäĒĢ£ ĒÖ£ņÜ® Ļ░ĆļŖźņä▒ņØä ĒÖĢņØĖĒĢśņśĆļŗż.
Abstract
The aim of this study was to analyze cells from human dental pulp tissue of impacted supernumerary teeth as stem cells with flow cytometry. Human dental pulp cells from 15 supernumerary teeth were identified their characteristics as stem cells by expression of mesenchymal stem cell markers through flow cytometry analysis at passage 3 and passage 10. Cluster of differentiation (CD) 73, CD 90, CD 34, CD 45 and STRO-1 cell surface markers were used to figure out characteristics of dental pulp stem cells from supernumerary teeth. At passage 3, the cell population showed positive expression of CD 73, CD90 and STRO-1, lacked expression of CD 34 and CD 45. At passage 10, CD 73, CD 90 and STRO-1 showed positive expression while CD 34 and CD 45 showed negative expression. This study indicated that dental pulp stem cells of supernumerary teeth had the properties of mesenchymal stem cells at both early and late passage. Impacted supernumerary teeth could be considered as a noble source of stem cells because of rapid growth and maintaining characteristics of stem cells until late passage.
Stem cells in human body have infinite potentials for cure or acceleration of healing in damaged tissue. Nevertheless, sources of stem cells to harvest out from human bodies are limited because of the difficulty of extracting many pure stem cells and the necessity of additional invasive procedure. Recently, it has been trying to find various sources of stem cell[1]. Embryonic stem cells have been treated as conventional stem cells for the last half century[2]. But the embryonic stem cells have limited supply and ethical issues. To replace the embryonic stem cells, studies on adult stem cells from dental tissues had been reported on various sources including dental pulp, extracted deciduous tooth, periodontal ligament, dental sac and apical follicle[3-7]. Most of stem cell researches in the dental field were related to the cells from wisdom teeth or deciduous teeth. However, the wisdom teeth have limitation of relatively late extraction time. Deciduous teeth exfoliate in an early age, but most of them has a physiologic root resorption and incomplete pulp tissue remnants. Impacted supernumerary teeth (SNT) can removed without damage of pulp tissue during surgery procedure at an early age[8].
The International Society for Cellular Therapy (ISCT) proposed minimal criteria for identifying mesenchymal stem cell (MSC) in 2006[9]. Previous studies of adult stem cells confirmed as MSCs by applying these criteria[10-12] while most studies used data obtained from a few or one donor.
The purpose of this study is to analysis characteristics of MSC of dental pulp from 15 impacted SNT.
Impacted supernumerary teeth in the anterior maxilla were extracted from 15 healthy patients who were between 6 and 9 years old. This study was approved by the Dankook university dental college institutional ethics committee (H-1506/006/001). Supernumerary teeth were kept in ╬▒-minimum essential medium (╬▒-MEM, Gibco) with 20% fetal bovine serum (FBS, Gibco), 100 U/mL penicillin, 100 ╬╝g/mL streptomycin (Gibco), 2 mM L-glutamine (Gibco) and 10 nM L-ascorbic acid (Sigma) immediately after extraction.
Supernumerary teeth were cut around at cementoenamel junction with dental disc until before exposing dental pulp. The crown and root were split along the groove to obtain dental pulp tissue with sterile dental file. Dental pulp tissue was chopped to fine slices less than 1 mm. Dental pulp cells were extracted using enzymatic digestion method with 3 mg/mL type I collagenase (Sigma-Aldrich) and 4 mg/mL Dispase (Sigma-Aldrich) under the shaking incubator at 37┬░C for 1 hour. Cells were filtered by 70 ╬╝m Falcon strainer (Corning) and cultured in ╬▒-MEM with 20% FBS, 100 U/mL penicillin, 100 ╬╝g/mL streptomycin, 2 mM L-glutamine. Culture solution was changed every 2 - 3 days, and floating particles were rinsed out. The cells were separated for next passage using trypsin-EDTA (Corning). Subculture until passage 10 was lasting for stemness comparison between cells of early and late passages.
Passage 3 and passage 10 cells were chosen to compare the expression of cell surface markers of early and late passage. Cells were counted to put same number of cells for each cell surface markers. 1 ├Ś 105 of the cells and 1 ╬╝g cell surface markers were leaved in refrigerator at 4┬░C for 1 hour. The surface markers used in this study were cluster of differentiation (CD) 73, CD 90, CD 34, CD 45 (BD Biosciences) and STRO-1 (R&D system). Inactive cell surface markers were removed and the fluorescent antibodies were used for florescence-activated cell sorting (FACS) analysis. The cell-antibody complexes were analyzed with BD FACSCalibur flow cytometry (BD Biosciences). The cells that were not reacted to any markers were used as control group.
Dental pulp cells were extracted from SNT of 15 patients; SNT 01 - SNT 15. Dental pulp cells were cultured to passage 10.
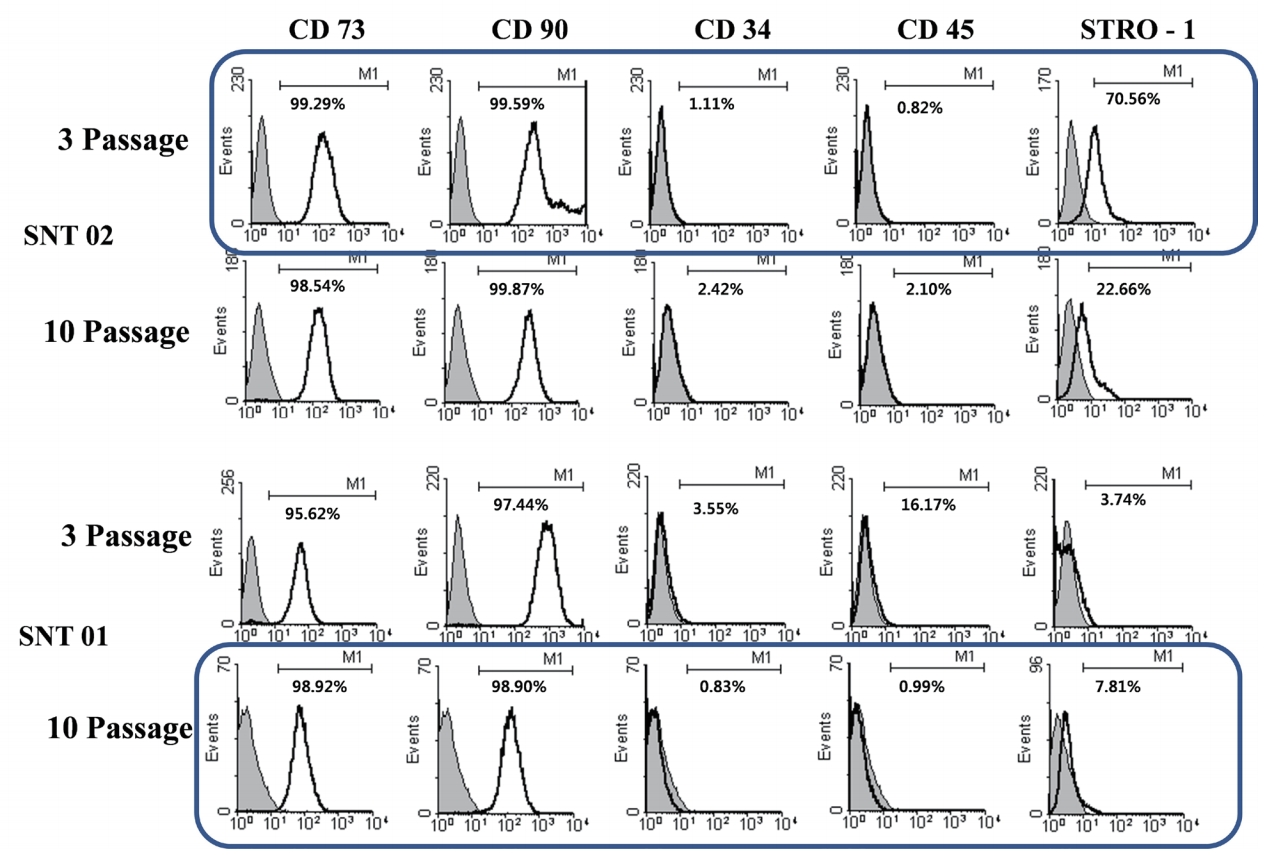
The expression of the cell surface markers in the control group and the experimental group was obtained at passage 3 and passage 10 of human dental pulp cells from SNT 01 and SNT 02 (Fig. 1). CD 73, CD 90, STRO-1 as MSC surface markers were highly expressed in the dental pulp cells from SNT, compared to only a small degree expression of CD 34, CD 45 as hematopoietic and endothelial markers. The expression of STRO-1 at passage 3 of SNT 02 and passage 10 of SNT 01 satisfy the criteria presented by ISCT.
Table 1 and 2 showed the expression of CD markers of the dental pulp cells from SNT at passage 3 and passage 10. CD 73 and CD 90 were positively expressed, while CD 34 and CD 45 were negatively expressed. STRO-1 was expressed. Most of dental pulp cells from SNT were satisfied with the criteria of ISCT.
Table 3 showed the mean value of expression of CD markers at passage 3 and passage 10. At passage 3, dental pulp cells from SNT showed positive expression of CD 73 (94.82%), CD 90 (98.86%) and STRO-1 (20.93%), negative expression of CD 34 (2.25%) and CD 45 (2.52%). At passage 10, dental pulp cells from SNT showed positive expression of CD 73 (96.62%), CD 90 (98.61%) and STRO-1 (35.62%), negative expression of CD 34 (3.86%) and CD 45 (4.14%). The expression of CD 73, CD90 and STRO-1 between two passages was not significantly different. There was a significant difference between two passage in expression of CD 34 and CD 45.
The average values of G-mean were calculated in flow cytometry results (Table 4).
The adult stem cells exist as undifferentiated cells in human organs[2,13,14]. Pittenger et al.[15] were classified the adult stem cells into MSCs and hematopoietic stem cells, and proved that the adult stem cells could be differentiated into osteoblasts, chondroblasts, myoblasts and nerve cells by controlling culture condition. The other studies reported the adult stem cells could be obtained from various tissues including bone marrow, pancreas, adipose, muscle, blood, hair follicles, skin and dental tissues[16].
In 2006, ISCT presented the minimal 3 criteria of MSCs[9]. First, they have characteristic of adhesion to plastic culture flask. Second, they must show positive expression of CD 105, CD 73 and CD 90, while as well as showing negative expression of CD 45, CD 34, CD 14 or CD 11b, CD 79alpha or CD 19 and HLA-DR. Thirdly, they should have a possibility of differentiation to osteoblasts, adipocytes and chondroblasts in vitro.
The aim of this study was to identify MSCs in 15 SNT with flow cytometry analysis. CD 73, CD 90 and STRO-1 were used as positive CD markers, and CD 34 and CD45 were used as negative CD markers.
STRO-1 is early mesenchymal stem cell marker, which was detected around blood vessels or nerve fascicles while not detected in size-sieved stem cells[12,17,18]. In 2007, Kolf et al .[19] reported STRO-1 was the best CD marker to identify mesenchymal stem cells. Gay et al .[20] reported that STRO-1 showed 27% expression in the cells from periodontal ligament of impacted wisdom teeth, and Park et al .[21] presented that STRO-1 showed 33.4% expression in the cells from periodontal ligament of supernumerary teeth. In this study, STRO-1 showed 20.93% expression at passage 3 and 35.62% at passage 10. The expression of the STRO-1 was even slightly increased in passage 10 comparing to passage 3. However, there was no significant difference between the two passages.
The expression of positive CD markers was not statistically different between passage 3 and passage 10 in flow cytometry analysis. This meant later passage of dental pulp cells from SNT still contained stemness and could have value of use as a source of stem cells. The expression of negative CD markers was a statistical significance between passage 3 and passage 10. According to ISCTŌĆÖs recommend, the negative CD marker should be detected lower, further study is required.
The dental pulp stem cells (DPSCs) of SNT from 15 donors showed characteristic of MSCs by flow cytometry using CD markers. The characteristic of MSCs was maintained until passage 10. However, FACS results alone are not enough to verify the stem cell function of cells. The differentiation to hard tissues or fat should also be examined. Also additional researches including real time qPCR, would be needed to confirm gene expression in follow-up studies.
The result indicated that DPSCs of impacted SNT had the characteristics of MSCs. Comparing passage 3 and passage 10 in flow cytometry analysis, the cell marker expressions were similar. There was no significantly difference between the positive CD marker values of passage 3 and passage 10. There was a significant difference in the negative CD marker values between the two passages. DPSCs maintained stemness until the late passage. It is confirmed that the possibility of using DPSCs of impacted SNT from young children as a donor of stem cells.
Fig┬Ā1.
Expression of cell surface markers in flow cytometry analysis of supernumerary tooth 01 and supernumerary tooth 02
at passage 3 and passage 10.
SNT : Supernumerary tooth, CD : Cluster of differentiation
Table┬Ā1.
Flow cytometry analysis of the mean percentile values of the expression of CD marker at passage 3
Table┬Ā2.
Flow cytometry analysis of the mean percentile values of the expression of CD marker at passage 10
Table┬Ā3.
The mean percentile values of flow cytometry analysis at passage 3 and passage 10
| Passage | Mean | SD | |
|---|---|---|---|
| CD 73 | 3 | 94.82 | 9.02 |
| 10 | 96.62 | 3.69 | |
| CD 90 | 3 | 98.86 | 1.17 |
| 10 | 98.61 | 1.18 | |
| CD 34 | 3 | 2.25*a | 1.90 |
| 10 | 3.86*a | 1.80 | |
| CD 45 | 3 | 2.52*b | 3.92 |
| 10 | 4.14*b | 2.79 | |
| STRO-1 | 3 | 20.93 | 17.69 |
| 10 | 35.62 | 27.69 |
References
1. Kim J : Characterization of differentiation of the supernumerary dental pulp stem cells toward the odontoblast by application period of additives. J Korean Acad Pediatr Dent, 42:312-318, 2015.


2. Evans MJ, Kaufman MH : Establishment in culture of pluripotential cells from mouse embryos. Nature, 292:154-156, 1981.



3. Gronthos S, Mankani M, Shi S, et al. : Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. 97:13625-13630, 2000.



4. Miura M, Gronthos S, Shi S, et al. : SHED: stem cells from human exfoliated deciduous teeth. 100:5807-5812, 2003.



5. Seo BM, Miura M, Shi S, et al. : Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet, 364:149-155, 2004.


6. Morsczeck C, Gotz W, Hoffmann KH, et al. : Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol, 24:155-165, 2005.


7. Sonoyama W, Liu Y, Shi S, et al. : Mesenchymal stem cellmediated functional tooth regeneration in swine. PLoS One, e79. 2006.



8. Kim J : Managing Complications Related to Multiple Supernumerary Teeth. J Korean Acad Pediatr Dent, 41:180-186, 2014.


9. Dominici M, Le Blanc K, Horwitz E, et al. : Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8:315-317, 2006.


10. Song JS, Kim SH, Jung HS, et al. : Characterization of stem cells obtained from the dental pulp and periodontal ligament of deciduous teeth. Tissue Eng Regen Med. 7:575-582, 2010.
11. Park JW, Song JS, Son HK, et al. : Effect of Storage Media and Duration on Pulpal Cell Viability in Exfoliated Deciduous Teeth. J Korean Acad Pediatr Dent, 41:1-7, 2014.

12. Kim D, Kim J, Roh S, et al. : A nanoscale ridge/groove pattern arrayed surface enhances adipogenic differentiation of human supernumerary tooth-derived dental pulp stem cells in vitro. Arch Oral Biol, 59:765-774, 2014.


13. Almeida-Porada G, Porada C, Zanjani ED : Adult stem cell plasticity and methods of detection. Rev Clin Exp Hematol, 5:26-41, 2001.


14. Temple S : Stem cell plasticity-building the brain of our dreams. Nat Rev Neurosci, 2:513-520, 2001.



15. Pittenger MF, Mackay AM, Marshak DR, et al. : Multilineage potential of adult human mesenchymal stem cells. Science, 284:143-147, 1999.


17. Chen SC, Marino V, Gronthos S, Bartold PM : Location of putative stem cells in human periodontal ligament. J Periodontal Res, 41:547-553, 2006.


18. Hung SC, Chen NJ, Lo WH, et al. : Isolation and characterization of size-sieved stem cells from human bone marrow. Stem Cells, 20:249-258, 2002.


20. Gay IC, Chen S, MacDougall M : Isolation and characterization of multipotent human periodontal ligament stem cells. Orthod Craniofac Res, 10:149-160, 2007.


21. Park JH, Song JS, Lee JH, et al. : Characteristics of stem cells derived from the periodontal ligament of supernumerary teeth. Tissue Eng Regen Med, 8:123-131, 2011.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print