Streptococcus mutans 바이오필름에 대한 에리스로신 매개 광역동 치료 시 potassium iodide의 효과
Effect of Potassium Iodide on Erythrosine-Mediated Photodynamic Therapy on Streptococcus Mutans Biofilms
Article information
Abstract
이 연구는 Streptococcus mutans 바이오필름에 대한 에리스로신 매개 광역동 치료에서 potassium iodide (KI)의 효과를 평가하는 In vitro 실험이다. S. mutans ATCC 25175를 배양하여 hydroxyapatite disk에 바이오필름을 형성하였다. 광감각제인 에리스로신을 20 μM, KI를 각각 10, 50, 100 mM로 희석하여 적용한 뒤 광역동 치료를 시행하였다. 생존한 세균의 수는 colony forming units (CFU)/mL로 산정하였으며 Bonferroni 사후 분석을 통해 그룹 간 차이의 유의성을 확인하였다. Confocal laser scanning microscopy (CLSM)을 이용하여 세포 생존율을 시각적으로 평가하였다. 실험 결과 KI를 적용한 후 광역동 치료를 시행한 실험군에서 KI의 농도의 관계 없이 현저한 CFU의 감소가 관찰되었다(p < 0.05). 또한 10 mM KI에 비해 100 mM KI를 적용한 실험군에서 유의한 CFU의 감소가 관찰되었다(p < 0.05). CLSM 관찰 시에도 동일한 결과를 확인하였다. KI는 모든 농도에서 S. mutans 바이오필름에 대한 에리스로신 매개 광역동 치료의 효과를 유의하게 향상시켰다. 이는 바이오필름 상태의 균주에 대한 광역동 치료의 낮은 감수성을 보완할 수 있지만, 추가적인 연구를 통한 최적의 임상 프로토콜 확립이 필요하다.
Trans Abstract
The aim of this in-vitro study is to evaluate the effect of potassium iodide (KI) on erythrosine-mediated photodynamic therapy (PDT) against Streptococcus mutans biofilms. S. mutans ATCC 25175 was cultured to form a biofilm on a hydroxyapatite disk. After diluting erythrosine to 20 μM and KI to 10, 50, and 100 mM, respectively, PDT was performed. The number of surviving bacteria was calculated as colony forming units (CFU)/mL and the statistical significance of the difference between groups was confirmed by Bonferroni post-hoc analysis. Cell viability was visually evaluated using confocal laser scanning microscopy (CLSM). As a result of the experiment, a significant decrease (p < 0.05) in CFU was observed in the experimental groups in which PDT was performed after applying KI regardless of the concentration of KI. In addition, a significant reduction (p < 0.05) in CFU was observed in the experimental group to which 100 mM KI was applied compared to 10 mM KI. The same results were confirmed when observing CLSM. KI significantly improved the efficacy of erythrosine-mediated PDT on S. mutans biofilms at all concentrations. This may compensate for the low sensitivity of PDT to biofilm-state bacteria strains, but it is necessary to establish an optimal clinical protocol through further research.
Introduction
Photodynamic therapy (PDT) exerts antimicrobial effects by inducing the generation of reactive oxygen species and active oxygen from photosensitizers that are activated following light irradiation [1]. PDT is used in clinical settings for treating cancers or skin diseases [2]. Moreover, PDT has been suggested as a promising alternative approach for caries prevention [3]. PDT is advantageous being a relatively conservative treatment strategy that is comfortable for patients, maintains normal microbial flora, and decreases the emergence of resistant bacterial strains [4,5].
Oral biofilm is a functional complex of various bacteria that plays an important role in the occurrence and progression of dental caries [6]. Microorganisms present in the biofilm exhibit a multi-matrix structure, and high resistance to antibacterial agents and the host immune system [7]. In addition, thick biofilms can limit the penetration of photosensitizers and restrict the light transmission [8]. Therefore, bacteria present in the biofilm possess relatively lower sensitivity to PDT compared to bacteria in the planktonic state.
Attempts have been made to overcome these limitations by using silver nanoparticle-based photosensitizers to improve their targeting capacity by bonding them to antibodies [9]. Recently, a novel approach to combining PDT with inorganic salts was reported [10]. Several inorganic salts can be used with photosensitizers, such as sodium azide, sodium bromide, and potassium bromide. However, previous studies have reported potassium iodide (KI) as being non-toxic and the most effective [10-12].
Among the various types of photosensitizers used for PDT in dental medicine, erythrosine is the main disclosing agent [13]. According to Wood et al. [13], erythrosine is the most effective photosensitizer in PDT for Streptococcus mutans biofilms, which is the main cariogenic bacteria. In the previous studies [14-16], the effect of PDT was evaluated using various photosensitizers along with KI; however, none of the studies have evaluated the effect of erythrosine to date.
Thus, this study aimed to evaluate the effect of KI at various concentrations on erythrosine-coupled PDT against S. mutans biofilms in-vitro .
Materials and Methods
1. Bacterial culture and culture conditions
S. mutans ATCC 25175 was inoculated in a brain heart infusion (BHI) broth (Becton, Dickinson and Company, Sparks, MD, USA), and cultured in a 5% CO2 incubator at 37℃ for 18 hours. Bacterial growth was monitored using a spectrophotometer (Smart Plus 2700; Young-woo instrument, Seoul, Korea) to determine the bacterial count. The bacterial suspension was diluted to a final concentration of 107 colony forming units (CFU)/mL using phosphate-buffered saline (PBS).
2. Biofilm formation
The hydroxyapatite (HA) disk (1.27 cm in diameter, Biosurface Technologies Corporation, Bozeman, MT, USA) was sealed using silicone impression material, except for the area needed for the treatment. After sterilizing with ethylene oxide gas, the HA disk was placed in a 12-well cell culture plate (SPL Life Sciences, Pocheon, Korea). Next, 1980 μL BHI broth and 20 μL S. mutans suspension were added to each well (final inoculation concentration: 105 CFU/mL) and the bacteria were cultured for 24 hours in an incubator set at 37°C and supplied with 5% CO2.
3. Photosensitizer and light source and potassium iodide (KI)
Erythrosine powder (Sigma-Aldrich, St Louis, MO, USA) was dissolved in PBS and used as a photosensitizer at a concentration of 20 μM. The erythrosine concentration was set based on a previous study by Choi et al. [17]. The solution was freshly prepared before the experiments, and exposure to light was minimized. A VALO light-emitting diode (LED) (Ultradent Products Inc., South Jordan, UT, USA) was used as a light source. The power intensity measured at an irradiation distance of 10 mm was 1200 mW/cm2, and the output was confirmed using a radiometer. The light irradiation distance was set, with consideration of the diameter of the LED curing beam and HA disk, to the minimum distance that is evenly irradiated throughout the disk.
Potassium iodide powder (Daejung, Siheung, Korea) was diluted to various concentrations (10, 50, and 100 mM) using sterile distilled water immediately before the experiment.
4. Experimental design
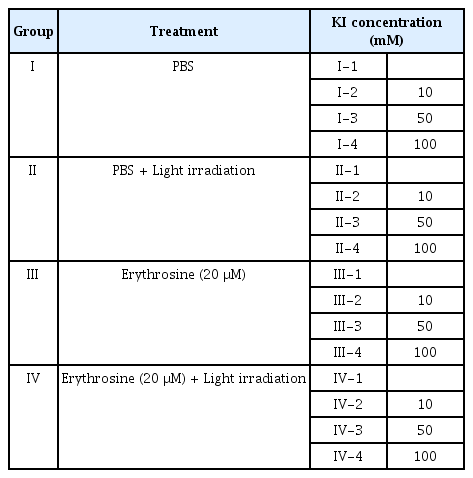
Four experimental groups were established as follows: Group I was treated with PBS without light irradiation; Group II was treated with PBS followed by light irradiation; Group III was treated with erythrosine without light irradiation; Group IV was treated with erythrosine followed by light irradiation. Subsequently, each group was subdivided into 4 groups based on whether or not KI was applied, and the different concentrations of KI used. The composition of the entire experimental group is shown in Table 1. For experiments, biofilm grown on the HA disk surface was treated with erythrosine, and the disks were incubated in a dark room for 3 minutes. Subsequently, light irradiation was performed at a distance of 10 mm for 15 seconds following KI application.
5. Colony forming units count
After treatment, each plate was washed using PBS to remove non-adherent bacteria. To turn the biofilm into a bacterial suspension, each well was sonicated twice for 10 seconds. The bacterial suspension was diluted using PBS and 50 μL aliquots were spread onto the blood agar plate (Hanil-KOMED, Seongnam, Korea) and incubated in a 5% CO2 incubator at 37°C for 72 hours. Each experiment was performed in duplicate.
6. Confocal laser scanning microscopy (CLSM)
CLSM was performed to evaluate PDT efficacy. Briefly, each specimen was washed with PBS and then stained using 200 μL LIVE/DEAD BacLight Bacterial Viability Kit Solution (Molecular Probes, Inc., Eugene, OR, USA) followed by incubation in dark. Stained biofilm was observed using LEICA TCS SP8 (Leica Microsystem, Wetzlar, Germany).
7. Statistical analysis
Data were analyzed using SPSS version 22.0 (SPSS Inc., Chicago, IL, USA). The normal distribution of the data was checked by performing the Shapiro-Wilk test. Bonferroni post-hoc analysis was performed to confirm the statistical significance of the difference between groups (p < 0.05).
Results
1. Colony forming units count
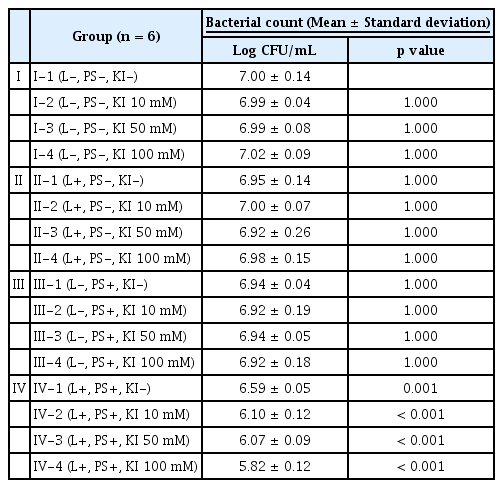
The CFU counts of all experimental groups are listed in Table 2. Compared to Group I-1, only 4 Group IV subgroups showed a statistically significant decrease in CFU counts (Fig. 1, p < 0.05). Within Group Ⅳ, 3 subgroups to which KI was applied (Group Ⅳ-2, 3, and 4) showed a significant decrease in CFU counts compared to the subgroup without KI treatment (Group Ⅳ-1, p < 0.05). Within the Group IV subgroups to which KI was applied, Group Ⅳ-4 showed a statistically significant decrease in CFU count compared to Group Ⅳ-2 (p < 0.05).
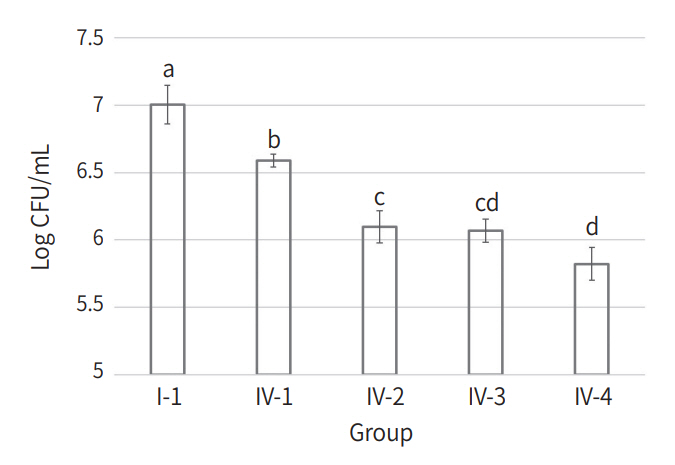
Mean and standard deviation of bacterial count. (1) Group I-1 (L-, PS-, KI-), (2) Group IV-1 (L+, PS+, KI-), (3) Group IV-2 (L+, PS+, KI 10 mM), (4) Group IV-3 (L+, PS+, KI 50 mM), (5) Group IV-4 (L+, PS+, KI 100 mM).
a,b,c,d: Means with the same letter are not significantly different by the Bonferroni’s post-hoc test.
2. Confocal laser scanning microscopy (CLSM)
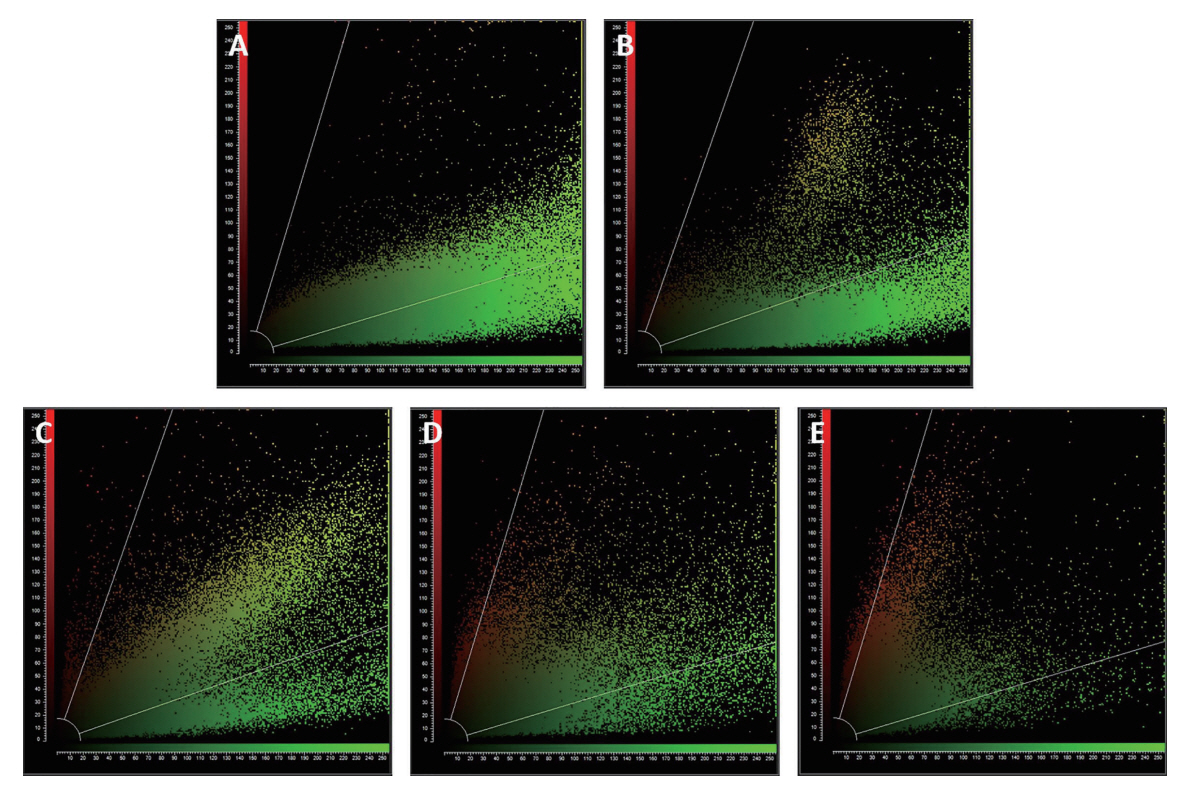
In CLSM observations, live bacteria emitted green fluorescence, whereas dead bacteria emitted red fluorescence. Images of Group I-1 and 4 experimental subgroups of Group Ⅳ are shown in Fig. 2. Group I-1 showed maximum green fluorescence, while Group Ⅳ showed increased red fluorescence. Even within Group Ⅳ, higher red fluorescence was observed in Group Ⅳ-2, 3, and 4 subgroups treated with KI, especially in Group Ⅳ-4. Fig. 3 shows the green/red fluorescence of CLSM images as a scatter plot based on Pearson’s correlation analysis.

Confocal laser scanning microscopy images. (A) Group I-1 (L-, PS-, KI-), (B) Group IV-1 (L+, PS+, KI-), (C) Group IV-2 (L+, PS+, KI 10 mM), (D) Group IV-3 (L+, PS+, KI 50 mM), (E) Group IV-4 (L+, PS+, KI 100 mM).
Discussion
KI is a FDA-approved thyroid blocking agent and is used for thyroid cancer prophylaxis [18]. As a thyroid blocking agent, the recommended dose of KI is 65 mg for children over 3 years, and 130 mg for adults per day. In this study, a markedly lower amount of KI was used (1mL of KI solution for each specimen) to ensure its biosafety when used clinically with PDT.
KI is present in Lugol’s solution, which is used as a disinfectant; therefore, KI was expected to exert an antibacterial effect independently. However, no significant effect was observed in this study, which was in line with earlier reports [14,19,20]. In aqueous solutions, KI reacts with singlet oxygen generated from activated photosensitizers and produces unstable peroxyiodide; it breaks down to produce stable antimicrobial species (free iodine and hydrogen peroxide) or short-lived reactive radicals that induce cell death [19,21]. Free iodine, a stable antimicrobial species, must reach a threshold concentration to exhibit antibacterial properties [19]. Stable antimicrobial species are mainly involved in gram-negative bacterial cell death, which has thin cell walls compared to other types of bacteria and is therefore easy to penetrate [19]. Alternately, reactive radicals are mainly involved in gram-positive bacterial cell death and exhibit antibacterial effects for a relatively short duration due to their short diffusion distance [21]. Based on these results, we speculate that the improvement of the antibacterial effect of PDT was mainly due to an increase in the production of reactive radicals. However, further research is needed to identify the exact mechanism.
PDT efficacy is affected by the distance and duration of light irradiation as well as the concentration and exposure duration of the photosensitizer [17]. Santos et al. [19] confirmed the potentiation effect of KI in PDT and reported that a comparable antibacterial effect was obtained with KI, even when a much lower concentration of photosensitizer was used. In the present study, significantly increased antimicrobial effects were observed at all KI concentrations in Group Ⅳ. Hence, KI can be a potential agent to enhance PDT efficacy to treat microorganisms present in biofilm.
Previous studies [15,20] have reported remarkable antibacterial effects with increasing KI concentration. In this study, a significant difference was observed between the antibacterial effects when KI was used at 10 mM and 100 mM concentrations.
This may be associated with increased antibacterial species and reactive radical production due to KI. The results of this study suggest that KI concentration may be critical for the clinical application of PDT with KI.
According to Choi et al. [17], the optimal concentration of erythrosine for PDT of S. mutans biofilm was 20 - 80 μM, and the minimum concentration was used in the present study. Furthermore, Kho et al. [22] reported that using erythrosine at clinically relevant concentrations exerted better antibacterial effects in PDT than those observed at a lower concentration. Therefore, applying clinically optimal concentrations of erythrosine and KI together is expected to increase the antibacterial effects of PDT. Consequently, further studies are required in this context to explore the ideal concentrations of these agents that can be used in combination in clinical settings.
In this study, the efficacy of KI in PDT on S. mutans biofilm, a representative cariogenic bacteria, was evaluated. However, in the actual clinical situation, cariogenic bacteria exist in a multispecies biofilm state. Multispecies biofilm is more resistant to PDT than monospecies biofilm due to differences in exopolysaccharide composition and interactions between several bacteria [23]. As the potential of KI in PDT of multispecies biofilm has not been studied yet, more studies in the future are needed to predict the actual treatment efficacy in the oral environment.
Conclusion
KI in combination with erythrosine significantly increased the PDT efficacy at all concentrations to treat S. mutans biofilms. KI has the potential to increase the efficacy of PDT for microorganisms present in biofilms. However, further studies are needed to establish optimal clinical protocols suitable for the oral environment.
Notes
Conflict of Interest
The authors have no potential conflicts of interest to disclose.