|
 |
| J Korean Acad Pediatr Dent > Volume 48(3); 2021 > Article |
|
초록
이 연구의 목적은 타이타늄-질소 코팅된 금속관(TiNCs)의 표면 경도와 스테인리스 스틸 금속관(SSCs)의 표면 경도를 비교하고, TiNCs의 부식 저항성 및 물리적 자극에 대한 금색 코팅의 안정성을 평가하는 것이다.
표면 경도 시험을 위해 10개의 TiNCs와 10개의 SSCs를 사용하였다. 각 금속관 종류당 총 30번의 경도를 측정하여 평균값을 구하고 비교하였다. 부식 저항성 시험을 위해 직사각형 형태의 TiNC와 SSC의 원재료(금속관으로 가공되기 전 판상 형태)를 준비하였다. 각 금 속관 종류 당 2회의 부식 시험을 시행하였다. 부식 용액에 용출된 금속 이온의 총 양을 유도결합 플라즈마 분광분석기로 측정하였다. 5개의 TiNCs로 반복적으로 가해지는 칫솔력에 대한 색조 안정성 시험을 시행하였다.
TiNCs의 평균 경도는 SSCs의 평균 경도보다 유의미하게 높았다. TiNCs와 SSCs의 원재료로부터 용출된 금속 이온의 총 합은 ISO 22674(재료의 무결성에 관한 기준) 기준치를 넘지 않음을 확인했다. TiNCs의 금색 코팅은 반복적인 물리적 자극에 대하여 주어진 시 간 동안 안정성을 가졌다.
Abstract
The purpose of this study was to compare surface hardness between titanium-nitride coated crowns (TiNCs) and stainless steel crowns (SSCs), and to evaluate the corrosion resistance and color sustainability of TiNCs.
Ten TiNCs and 10 SSCs were used for the hardness test. Measurement was performed 30 times for each type of crowns, and the mean values were compared. Metallic raw material plates (before being processed into crowns) of TiNCs and SSCs were prepared for the corrosion resistance test. The total amounts of metal ion releases in the test solution were detected by inductively coupled plasma-optical emission spectrometry. Five TiNCs were subjected to the color sustainability test by applying repetitive brushing forces.
The mean hardness values of TiNC group and SSC group were 395.53 ± 105.90 Hv and 278.70 ± 31.45 Hv respectively. Hardness of TiNCs were significantly higher than that of SSCs. The total amounts of metal ion releases from the materials of TiNCs and SSCs satisfied the criterion in International Organization for Standardization 22674. The results mean that TiNCs and SSCs were not harmful in an acidic environment. The golden coating was stable against the repetitive physical stimulations for a given period time.
Stainless steel crowns (SSCs), first reported in 1950 by Engel and Humphrey, have been chosen to restore damaged teeth due to severe caries, traumatic fractures, or developmental defects[1]. SSCs are primarily recommended where pulpotomy or pulpectomy is done presuming there is less leakage in rehabilitated teeth in comparison to those restored with amalgam[2,3]. Great durability, low occurrence of secondary caries, favorable retention and convenience in use are also advantages of SSCs’[4].
Full coverage crowns for primary molars have come in different forms at market on demand of esthetic appreciation and mechanical properties: SSCs, resin-veneered crowns (RVCs), and zirconia crowns[5]. SSCs and RVCs are basically made of metal alloys which are frequently employed in the dental field: restorations, orthodontic appliances, and endodontic instruments. The surface of these dental instruments may be coated in order to resist corrosion and reduce bacterial attachment in an intraoral environment[6,7].
Recently, a new full coverage crown of which surface is coated with titanium-nitride (TiN) has been introduced: titaniumnitride coated crown (TiNC). TiN-coating makes the crown look golden and yellow. The coating has been used in medical field for patients with heart failure and for pacemaker leads[8,9]. Dental implants also have TiN-coating for its excellence in biological and mechanical properties and the esthetic aspect of the “golden color”[10-12]. The coating may play a role in strengthening hardness of the surface[13].
Hardness of restoration materials is an important factor in clinical application. If hardness is too weak, it is easy to cause fracture and perforation[14,15]. In Roberts et al .[16] study, the perforation rate of SSCs was approximately 2%. Hardness of TiNCs was expected to be more improved than that of conventional SSCs.
As one kind of dental material, a degree of safeness of TiNCs in an acidic environment needs to be verified. Intra oral condition becomes acidic when food is consumed and the acidified oral environment makes tooth decay more prone[4]. There is an international standard regarding dental material: the International Organization for Standardization (ISO) 22674 for dentistry - metallic materials for fixed and removable restorations and appliances. A criterion is specified in ISO 22674: 2016, 5.7 Corrosion resistance for material integrity[17]. Safeness of TiNCs in an acidic setting has not yet been studied.
This study was conducted to compare surface hardness of TiNCs and that of SSCs, to evaluate corrosion resistance of TiNCs, and to assess color sustainability of the coating of TiNCs from physical stimulations.
TiNCs (Kids Golden Crown, Shinhung, Seoul, Korea) and SSCs (Kids Crown, Shinhung, Seoul, Korea) for maxillary right primary second molars (FDI tooth numbering system 55) sized 5 were used (Fig. 1). Chemical compositions of the raw material (stainless steel) for TiNCs and SSCs and the types of coating were listed in Table 1. Sizes of a TiNC and an SSC were the same when measured by a vernier calipers (Mitutoyo, Tokyo, Japan). The thicknesses of TiN-coating (of TiNCs) and SiO2 coating (of SSCs) were 4 - 6 μm and 1 μm respectively.
Ten TiNCs and 10 SSCs were used for the hardness test. Crowns were embedded in self-cured acrylic resin (Pace technologies, Arizona, USA) with the occlusal surface up. For convenience, crowns were buried in groups of 3, 3, and 4 for each crown type. A cylinder shape of the specimens was sized 30.0 mm in diameter and 18.0 mm in height. The surface was ground by sandpaper grip 400 (Buehler, Illinois, USA) until the occlusal surface was visible. Then the surface was ground by sandpaper grip 600 and 800 sequentially. At least 3 parts apart from each other were exposed on the occlusal surface (Fig. 2). Each exposed part was at least 2.0 × 2.0 mm. The prepared surface was finally polished with 3 μm diamond suspension (Buehler, Illinois, USA).
For one test, 2 rectangular plate specimens with dimensions of approximately 33.0 × 11.0 mm (approximate surface area 3.7 cm2) from the flat materials of TiNC and SSC were prepared for the corrosion resistance test (Fig. 3). Totally, 4 plates from TiNC material and 4 plates from SSC material were prepared for twice testing. The acidic test solution was prepared according to ISO 22674: 2016, 8.9.4 Test solution[17].
Five TiNCs were prepared for the color sustainability test. Each crown was fixed with glass ionomer cement (3M ESPE, Seefeld, Germany) on the applicator so that the smooth surface of the crown was applied. As a source of physical stimulation, a tooth brush (Shinhung, Seoul, Korea) was held firmly with a cable tie on the moving part of the abrasion test machine (Shinhung, Seoul, Korea) (Fig. 4).
A total of 20 crowns (10 TiNCs, 10 SSCs) were subjected to the Vickers hardness tester (Shimadzu, Tokyo, Japan). The load at 1.961 N was applied to the occlusal surfaces in an occlusogingival direction for 10 seconds. Measurement was performed 3 times for each crown at least 2.0 mm apart from each indented point. Thirty hardness values from TiNCs and 30 values from SSCs were collected. An average value for each group was calculated from the data.
Specific qualitative and quantitative requirements for freedom from biological hazard were not included in ISO 22674. The test was conducted twice for each metallic material.
Rectangular specimens in pairs were placed in a separate laboratory dish, and the acidic solution was filled in a ratio of 1 mL/cm2 of the surface area of the specimens. The containers were sealed and maintained at 36.5°C for 7 days. The test solution from which the metal ions were eluted was quantitatively analyzed by inductively coupled plasma-optical emission spectrometry (ICP-OES, PerkinElmer Inc., California, USA). Fe, Cr, Mn, Ni, Si, and Ti were set to be analyzed. Fe, Cr and Mn are classified as heavy metals, and Ni is allegedly known as an allergen. Si is an element composing of raw materials for stainless steel and the coating of SSCs. Ti is a constituent material of the coating of TiNCs. The total metal ion releases from the 2 metallic materials were recorded.
Five TiNCs were subjected to the color sustainability test. The test was conducted using the abrasion test machine. The machine was designed to assess how sustainable the golden TiN-coating was. It was equipped with a specially designed tooth brush on the moving table that moved over the smooth surface (distal surface) of the subjected crown back and forth in a parallel way (Fig. 5). Brushing forces of 1 N were repetitively given through the movement of the tooth brush. The screen of the machine showed a total count of the brushing movements. A speed of the tooth brush was adjusted to the maximum speed of 4 times/sec. The test was conducted for 30 days, 6 hours a day.
The means of TiNC group and SSC group were 395.53 ± 105.90 Hv and 278.70 ± 31.45 Hv respectively (Table 2). Hardness of TiNCs was higher than that of SSCs. There was a significant difference between the two groups (p < 0.0001).
The total metal ion releases were 12.988 μg/cm2, 15.951 μg/cm2 from the TiNC material and 6.370 μg/cm2, 6.909 μg/cm2 from the SSC material. The amount of each metal ion release was shown in Table 3.
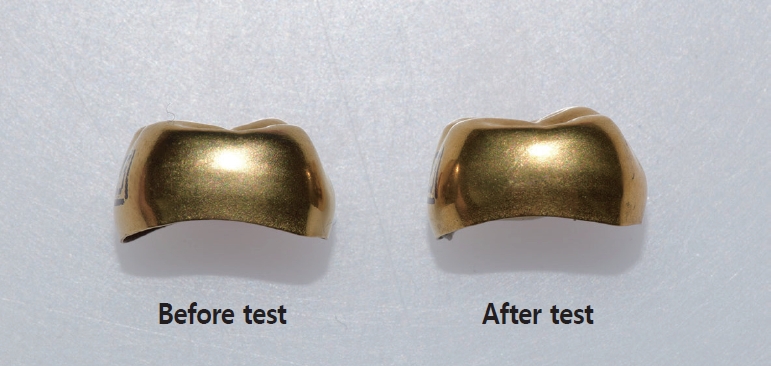
The golden coating of all the 5 TiNCs were intact with the naked eye. The golden TiN-coating did not come off until the count of brushing movements hit 2,592,000 (Fig. 6).
Full coverage crowns including SSCs have a thin coating on the surface. Jonsson and Hogmark[18] said that coatings had been increasingly used to improve mechanical or electrical properties, corrosion resistance or for a decorative purpose. According to the manufacturer, TiNCs are made by putting TiN-coating instead of SiO2 coating which is overlaid on SSCs. TiNCs have a thicker outer layer (4 - 6 μm) than SSCs’ (1 μm). In this study, the hardness of TiNCs was significantly higher than that of SSCs. This might be due to the thicker thickness of TiN-coating or the hardness of the coating itself. In Attar’ s[13] study, the increase in ceramic coating thickness resulted in an increase in surface hardness of the substrate.
Means of enamel micro-hardness of primary molars were 367.7 - 396.4 Hv[19]. In vitro, mean micro-hardness of SSCs was 161.58 Hv[20]. If hardness of metal crowns is higher than that of tooth enamel, the tooth might wear off. If vice versa, the crown might be worn out and perforated. The average occlusal bite force of primary molars was 146.06 N, as a reference value[21]. It is presumed that desirable hardness for preformed crowns should be similar to or slightly lower than that of tooth enamel.
The standard deviation obtained in this study appeared high. One of the possible reasons might be that the surfaces of specimens were not evenly treated during the preparation. Part of the surface coating might have been peeled off during the grinding process to flatten the subjected occlusal surface. One possible solution would be to use a relatively flat smooth surface (buccal or palatal) as an experimental surface.
Regarding the thickness of coating, the coating thickness of crowns would affect the elasticity felt by the operator during crown trials. Future studies are needed to test comparing the elasticity of TiNCs and SSCs during crown trials.
The purpose of conducting the corrosion resistance test was to see the total amounts of metal ion releases. An amount of each metal ion release was not of interest. The obtained data of metal ion releases satisfied the criterion in ISO 22674. A statistical significance of the test was not considered. Limitations of this test included that its small sample size and absence of comparison data between the 2 groups. In future studies, statistical significance between the 2 groups may be derived from a larger number of samples and more times of testing.
Since instruments and appliances with TiN-coating including TiNCs are currently in use at clinics, it is assumed that there are practically no problems with biological hazard. Although Ni and Cr were reportedly allergy-inducing agents, there were few cases of allergic reactions to Ni or Cr containing dental materials[22]. Wiltshire et al .[22] claimed that there was little evidence to implicate that these metals played a role in outbreaking of adverse reactions. The authors continued to say that it might be concluded that Ni and Cr were generally safe to use in dentistry. Mn is classified as heavy metal, but its toxicity as a dental material has not been reported yet. Clinicians nonetheless should always be alert about possibility of any metal-inducing allergies[22].
One differentiating factor of TiNCs is their unique color compared to SSCs. TiNCs are chosen over SSCs by parents and children because of TiNCs’ shiny golden color benefits. When a person opens his or her mouth wide or smiles, posterior teeth regions are visible. In the color sustainability test, experimental surfaces were examined by the naked eye. Whether or not the golden coating of a TiNC placed in the oral cavity has been peeled off could be determined visually even at a certain distance. In this study, it needed to be confirmed whether the golden coating was peeled off or not in the eyes of the parents and children. Examining color changes of the coating of TiNCs and SSCs with a microscope or spectrophotometer instead of naked eye might have been a more scientific and qualitative approach.
Messer and Levering[23] reported that placed SSCs were successfully used for 68.2 months for those who had the restorative crowns set younger than 4 years and 36.1 months for those who had them 4 years and older. One would use an SSC for about 3 to 6 years until the tooth exfoliates. American dental association recommends brushing teeth twice a day[24]. Considering twice or three times of tooth brushing a day, the number of repetitive brushing movements per tooth surface would add up to about 1100 per year and 7000 for 6 years if calculated generously. The color sustainability test was run for 30 days, 6 hours a day, and the count of bushing movements was 2,592,000. The counting was thought to be sufficient to see if the golden coating was worn off or not until a crowned tooth spontaneously fell off. In this study, it was confirmed that the golden color was not peeled off easily. If the golden color was quickly peeled off, there would have been no reason to choose higher priced TiNCs. Therefore, sustainability of the golden color is one of the crucial factors for choosing TiNCs.
Cases in which the golden coating may come off should also be kept in mind by the clinicians. If bruxism or clenching habit is present, greater occlusal forces for a longer period time are applied on crown surfaces. Maximum occlusal bite force when clenching reached over 400 N[25]. Considering of the normal occlusal force, approximately 150 N, repeated, unnecessary and excessive occlusal force leads to fatigue accumulation, which results in peeling of the coating and eventually perforation[21].
The introduction of TiNCs is thought to be meaningful in that it has broadened the range of options for posterior teeth restoration. TiNCs could be a better choice for children who have oral habits by virtue of TiNCs’ comparatively strong hardness. Biohazard over metal materials for pediatric dentistry is often questioned by parents at dental clinics. It could be explained that the materials of TiNCs and SSCs are not harmful based on the results of the corrosion resistance test. Before choosing TiNCs, parents would ask if the golden coating would peel off easily. It would be answered that the coating was sustainable and did not come off readily. TiNCs may content potential patients and their parents who have demand for the golden shiny color.
As TiNCs are newly introduced in pediatric dental clinic, not many studies have been researched so far. Topics could be further studied such as failure rate and the amount of tooth reduction compared to other full coverage crowns. In addition to the hardness test carried out in this study, testing TiNCs' wear strength on the opposing tooth using stylus would be a good attempt in prospective studies.
Mean hardness of TiNCs was significantly higher than that of SSCs. The total metal ion releases from the metallic materials of TiNCs and SSCs satisfied the criterion in ISO 22674 in the corrosion resistance test. The golden TiN-coating was sufficiently stable against repetitive brushing forces for a certain period of time.
Fig 3.
Rectangular plate specimens of 2 materials for corrosion resistance test (each sized 33.0 × 11.0 mm).

Table 1.
Chemical compositions of the raw material for titaniumnitride coated crowns and stainless steel crowns, and the types of coating
Table 2.
Means and standard deviations of surface hardness
| Group | LOT No. | Mean ± SD (Hv) | p value |
|---|---|---|---|
| TiNCs | RG190108 | 395.53 ± 105.90 | < 0.0001 |
| SSCs | RS192123 | 278.70 ± 31.45 |
Table 3.
Metal ion releases from the metallic materials of titanium-nitride coated crowns and stainless steel crowns in corrosion resistance test
References
1. Berg JH : The continuum of restorative materials in pediatric dentistry-a review for the clinician. Pediatr Dent, 20:93-100, 1998.

2. Croll TP, Killian CM : Zinc oxide-eugenol pulpotomy and stainless steel crown restoration of a primary molar. Quintessence Int, 23:383-388, 1992.

3. Holan G, Fuks AB, Ketlz N : Success rate of formocresol pulpotomy in primary molars restored with stainless steel crown vs amalgam. Pediatr Dent, 24:212-216, 2002.

4. Kim KC, Kim S, Choi YC, et al. : Pediatric Dentistry. 5th ed. Dental Wisdom, Seoul, 274-277, 354. 2014.
5. Fieldman BS, Cohen MM : A simple efficient method for utilizing the stainless steel crown. ASDC J Dent Child, 46:464-469, 1979.

6. Puckett SD, Taylor E, Raimondo T, Webster TJ : The relationship between the nanostructure of titanium surfaces and bacterial attachment. Biomaterials, 31:706-713, 2010.


7. Sterzenbach T, Helbig R, Hannig C, Hannig M : Bioadhesion in the oral cavity and approaches for biofilm management by surface modifications. Clin Oral Invest, 24:4237-4260, 2020.

8. Schaldach M, Hubmann M, Hardt R, Weikl A : Titanium nitride cardiac pacemaker electrodes. Biomed Tech (Berl), 34:185-190, 1989.

9. Sin DC, Kei HL, Miao X : Surface coatings for ventricular assist devices. Expert Rev Med Devices, 6:51-60, 2009.


10. Mezger PR, Creugers NH : Titanium nitride coatings in clinical dentistry. J Dent, 20:342-344, 1992.


11. Wisbey A, Gregson PJ, Tuke M : Application of PVD TiN coating to Co-Cr-Mo based surgical implants. Biomaterials, 8:477-480, 1987.


12. van Hove RP, Sierevelt IN, van Royen BJ, Nolte PA : Titanium-Nitride Coating of Orthopaedic Implants: A Review of the Literature. BioMed Res Int, 2015:1-9, 2015.

13. Attar F : Hardness evaluation of thin ceramic coatings on tool steel. Surf Coat Technol, 78:78-86, 1996.

14. O’Quigley DGF, Luyckx S, James MN : New results on the relationship between hardness and fracturetoughness of WC-Co hard metal. Mater Sci Eng, 209:228-230, 1996.

15. Behrmann LA, Elbel JL : Effect of Perforations on Fracture Initiation. J Pet Technol, 43:608-615, 1991.

16. Roberts JF, Attari N, Sherriff M : The survival of resin modified glass ionomer and stainless steel crown restorations in primary molars, placed in a specialist paediatric dental practice. Br Dent J, 198:427-431, 2005.


17. International Organization for Standardization : International Standard ISO 22674. 2nd ed. ISO, 8-22, 2016.
19. Bahrololoomi Z, Lotfian M : Effect of Diode Laser Irradiation Combined with Topical Fluoride on Enamel Microhardness of Primary Teeth. J Dent (Tehran), 12:85-89, 2015.


20. Guitierrez MN, Lopez SA : In vitro evaluation of microhardness of amalgam and stainless-steel crowns. Rev Odotopediatr Latinoam, 8:123-130, 2018.
21. Owais AI, Al-Battah AH, Abu Alhaija ES : Changes in occlusal bite force following placement of preformed metal crowns on primary molars in 4-6 years old children: a 6 months' follow-up pilot study. Eur Arch Paediatr Dent, 20:9-14, 2019.


22. Wiltshire WA, Ferreira MR, Ligthelm AJ : Allergies to dental materials. Quintessence Int, 27:513-520, 1996.

23. Messer LB, Levering NJ : The durability of primary molar restorations: II. Observations and predictions of success of stainless steel crowns. Pediatr Dent, 10:81-85, 1988.

24. American Dental Association : Brushing Your Teeth. Available from URL: https://www.mouthhealthy.org/en/az-topics/b/brushing-your-teeth (Accessed on September 16, 2019)
- TOOLS







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print